In-depth topics
Update on airborne transmission of COVID-19: evidence and implications for precautions
Since the beginning of the COVID-19 pandemic, our understanding of transmission of the causative virus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has evolved. Public health organizations, including the World Health Organization (WHO) and the Centers for Disease Control and Prevention (CDC) have emphasized that transmission of SARS-CoV-2, like many other respiratory viruses, is driven by respiratory droplets. The public health and social measures (PHSM) recommended to reduce transmission of SARS-CoV-2 outside health care settings include hand-washing, physical distancing and wearing face coverings. These recommendations take cues from infection prevention and control (IPC) guidelines for health care settings for the prevention of respiratory infections spread through droplet transmission. Under IPC guidelines, there are several tiers of infection control. These include standard precautions (basic measures such as hand-washing that are used for all patients to generally prevent the spread of infectious diseases), and transmission-based precautions that are adopted if a specific pathogen is suspected or diagnosed. There are a bundle of interventions for each possible transmission route, which includes detection of those who may be infected, placement of potentially infectious patients in physically separate areas (e.g., in rooms with specific engineering controls that reduce the risk of pathogen exposure) and use of specific personal protective equipment (PPE) by health care workers and visitors. Respiratory pathogens are transmitted when mucus or saliva containing live infectious pathogens come into contact with mucous membranes (e.g., membranes in the eyes, mouth, lungs or nose) of a susceptible person. This occurs when an infectious person exhales droplets during activities such as speaking, singing, sneezing or coughing. In the first half of the 20th century, tuberculosis researchers proposed classification of respiratory droplets into smaller and larger sizes. Although increasingly sophisticated research methods have challenged this dichotomy, and there is evidence that exhalations generate multiphase turbulent clouds that carry droplets across a continuum of sizes, this droplet size classification is still used today.
“Airborne transmission” refers to transmission in small droplets (variably referred to as “aerosols” or “droplet nuclei”) that are either expelled from the respiratory tract or left behind when larger droplets evaporate and may stay suspended in the air for hours. Aerosols may be dispersed over long distances by air currents and then inhaled by others who have not been near, or even in the same room as, the infectious individual. Precautions to prevent airborne transmission include the use of face coverings over the mouths and noses of people with infection, special ventilation systems that collect and remove potentially infectious airborne particles, as well as the use of specialized masks such as N95 respirators that are designed to filter out tiny particles. Pathogens considered to be transmitted by the airborne route include Mycobacterium tuberculosis (the causative agent of tuberculosis), rubeola virus (the causative agent of measles) and varicella-zoster virus (the causative agent of chickenpox and shingles). All are known to infect people across longer distance ranges and for all three pathogens, the number of people typically infected by each infectious person (the basic reproduction number, or “R naught,” abbreviated R0) is extremely high: in the 10-20 range, approximately 4-8 times higher than the estimated R0 for SARS-CoV-2. “Droplet transmission” refers to transmission via larger droplets that travel short distances before evaporating or falling out of the air, potentially contaminating surfaces. Precautions to prevent droplet transmission include the use of face masks (sometimes called surgical masks) to block droplet contact with mucous membranes. There are several respiratory viruses considered to be primarily transmitted via droplets, despite some evidence that airborne transmission can also occur. These include influenza and respiratory syncytial virus, a common virus that causes cold-like symptoms. For these viruses, long-range transmission has rarely been observed and the R0 is much lower. The size cut-off between aerosols and droplets is variable; the WHO and the CDC consider aerosols to be less than 5 micrometers and droplets to be over 5 micrometers.
Many variables influence the transmission dynamics of respiratory viruses, including the amount of virus sufficient to cause an infection, the susceptibility of the exposed person, the potential of the virus to come into contact with a person’s mucous membranes and the viability of that virus (whether it is alive and able to cause infection). Many environmental variables also influence the spread and trajectory of exhaled droplets through the air. To discern the precise relative importance of different transmission modes, it would be necessary to implement human challenge trials, in which humans are purposefully exposed to a pathogen in controlled environments. Since such trials are often ethically impossible to perform, and have not been performed for COVID-19, much of the evidence that guides infection control recommendations comes from epidemiologic studies on disease transmission events.
Knowledge of how SARS-CoV-2 is transmitted is informed in part by disease outbreaks. Superspreader events provide important, albeit indirect, evidence of transmission mode. The well-publicized COVID-19 outbreak linked to an air-conditioned restaurant in Guangzhou, China, may be illustrative of droplet transmission given the pattern of air flow induced by the air conditioner and the number of people in the restaurant who did not get infected. However, a non-peer reviewed analysis of the same superspreader event suggested there was evidence of potential airborne transmission. A 2.5 hour choir practice in Washington State, during which one person with COVID-19 infected 52 others, provided evidence that indoor, close-range interactions facilitate transmission. Although this superspreader event was likely fueled by droplet transmission, the possibility of some degree of airborne transmission, potentially fueled by the act of singing, could not be ruled out. The same is true of a COVID-19 cluster in South Korea associated with fitness dance classes; short-range aerosol transmission, particularly in crowded and inadequately ventilated spaces over a prolonged period of time, could have occurred. As with many complex biological processes, there is likely a continuum of transmission risk across a number of variables, including environmental conditions (indoor versus outdoor), duration of exposure and potentially, characteristics of the index (source) case, that increase or decrease the risk of transmission. A recent systematic review found evidence to suggest that SARS-CoV-2 may travel beyond 6 feet from an infectious person. Air sampling studies have provided evidence that SARS-CoV-2 may be airborne. However, the methods used in the different studies have varied to the extent that definitive conclusions are difficult to draw, and few studies have attempted to determine whether air samples contain viable virus (potentially able to cause an infection) or only viral genetic material.
In their letter to the WHO, authors and signatories expressed concern that airborne transmission may play a significant role in the epidemiology of COVID-19, to the point that recommendations on methods to reduce disease transmission may need to be revised. There are several examples of pathogens, including the virus that causes severe acute respiratory syndrome (SARS), for which infection control guidelines were amended as evidence of transmission mode accumulated. However, undertaking more stringent (airborne) precautions for respiratory pathogens when the possibility of airborne transmission cannot be ruled out may be difficult due to fiscal, space, and human resource considerations as well as the availability of equipment and materials. Per the CDC, “N95 or higher level respirators are routinely recommended for emerging pathogens like SARS CoV-2, which have the potential for transmission via small particles…CDC recommendations acknowledge the current challenges with limited supplies of N95s and other respirators….facilities that do not have sufficient supplies of N95s and other respirators for all patient care should prioritize their use for activities and procedures that pose high risks….” There is debate about what interventions are most important to protect the public’s health when data are limited. The letter to the WHO argues that public health guidelines should follow the precautionary principle and address any and all possible pathways to curb pandemic spread, but managing the opportunity costs of such recommendations can be challenging.
In response to the open letter, the WHO released an overview of evidence on transmission of SARS-CoV-2 on July 9. The review concluded that SARS-CoV-2 is primarily spread through contact and respiratory droplets, but that airborne transmission may occur under some circumstances, such as during certain medical procedures conducted in health care settings. The review emphasized that “limiting close contact between infected people and others is central to breaking chains of transmission of the virus causing COVID-19.” There was no indication that specific recommendations to reduce transmission will be imminently changed. As the pandemic progresses, new scientific information will become available and updates to recommendations may be warranted. Ultimately, basic concepts in respiratory disease transmission may be revisited. For now, available evidence definitively supports the importance of everyone practicing the “3 W’s for a safer reopening:” Wear a mask, Wash your hands and Watch your distance. In addition, everyone should “Avoid the Three Cs:” Crowded places, Close-contact settings and Confined and enclosed spaces.
Expanding social networks safely through bubbles
In March 2020, as the novel coronavirus SARS-CoV-2 was spreading globally, many countries instituted strict public health and social measures (PHSMs) to limit spread and control the COVID-19 pandemic. These PHSMs often included lockdowns and stay-at-home orders designed to flatten the growth in new cases, allowing healthcare systems to build capacity and avoid being overwhelmed. Prolonged lockdowns can have significant negative social and economic impacts, and although effective in the short term, need to be transitioned to a more sustainable model. One such model allows people and households to extend their networks by introducing bubbles.
Bubbles were first suggested as a strategy to ease through COVID-19 lockdowns in New Zealand. A bubble refers to an entity, such as all the members of a household, who are regularly in contact with one another but separated from physical interaction with others. The general principle behind bubbles is that by carefully extending social and physical contact beyond one household bubble or unit to include another household bubble or unit (e.g., a neighbor, close friend, or extended family), people will be able to ease out of the isolation of lockdowns while keeping the risk of transmitting COVID-19 low. Those inside the newly extended bubble do not need to physically distance from one another, but must continue to physically distance from everyone else. Rather than go directly from a state of complete lockdown and isolation to limitless contact with others allowing for potentially uncontrollable opportunities for virus transmission, a double-bubble can serve as a “middle-ground approach that expands social interaction and contains risk by limiting exposure,” according to an epidemiologist from American University.
There is no single definition of what a bubble constitutes, and individuals and families have to factor different priorities when considering this approach. What all bubbles have in common is that they allow for increased interpersonal contact while minimizing the risk of transmission by clustering contacts. To work properly, bubbles must have rules, and those agreeing to join bubbles must agree on these rules. Each household on its own is considered a bubble. Joining bubbles can create a larger or extended bubble. A single adult living in a one-person household, for example, may choose to form an exclusive extended bubble with the member or members of another household. However, in most circumstances, multiple single adults living together cannot each choose a different second household to form a bubble with. Families with children may want to choose an extended bubble to join based on their children’s playmates. Those with elderly or high-risk household members may choose not to extend their bubbles at all. If anyone in the bubble develops symptoms or becomes ill, all members of the extended bubble must quarantine. Some countries, such as the U.K., are using bubbles as part of their national strategy, and offer residents formal guidelines on do’s and dont’s. In Marin County, California, “social bubbles” are part of official guidance for reopening, and though they do not allow for interaction indoors, they do allow for a “stable group of not more than 12 individuals” to attend outdoor social events together without mixing with other social bubbles. A similar concept is used by Alameda County in California.
Though bubbles have not been studied directly, research using models and existing knowledge about COVID-19 transmission has shed some light on how bubbles may be part of the solution to mitigate the negative aspects of social isolation and lockdowns. One preprint study showed that extending contacts by joining two household bubbles can result in a significantly lower mortality risk from COVID-19 than extending contacts without bubbles. The fewer people involved in the extended bubble, the lower the risk for infection transmission. Extending bubbles with people who share similar characteristics such as age and geographic location, was also shown to reduce transmission risk.
Other contexts in which the word “bubble” has emerged recently with relation to COVID-19 are athletics and travel. In athletics, the U.S. National Basketball Association (NBA) has chosen to move the entire league to Disney World, which will serve as a “pandemic isolation zone.” Players and staff will be separated from the outside in this bubble that serves to keep them safe, but also allows play and league activities to continue under regular monitoring and testing by doctors and health care providers. This closed network is serving as a unique and relatively controlled environment where researchers can work with the league to further COVID-19 science, especially around testing and virus spread. In travel, bubbles would serve to extend units but on a much larger scale; extending travel between two countries or among a small network of countries. One such network is under consideration for Australia, New Zealand and Fiji, an island nation highly dependent on its trade and tourism partners. Part of what would support the development of this travel bubble is the relative success that all three of the countries have enjoyed in addressing the pandemic overall, although the evolving situation in Australia may affect these plans.
The end of the COVID-19 pandemic is not yet in sight. As some parts of the world begin to ease out of the worst, others are just beginning to experience the greatest impact from the virus, with stressed health care systems and increasing COVID-19 cases, hospitalizations and deaths. What appears certain is that some degree of PHSM implementation is here to stay for the foreseeable future. By using data for action, taking individual and population risk into account, and encouraging careful and educated choices about the expansion of social networks, bubbles may be one part of a COVID-19 lockdown exit-strategy that can allow people to interact with one another more safely. The larger the number of people in a bubble, the greater the likelihood that one member will become infected from outside the bubble; all members of the bubble must be strictly isolated from potential infection to keep the bubble secure. Outside of bubbles, extended or not, people need to continue to wash their hands, wear masks and watch their distance to pave a way forward through the pandemic.
Weekly Research Highlights
OpenSAFELY: Factors Associated with COVID-19 Death in 17 Million Patients
(Nature, Accelerated preview, July 8)
-
The researchers used a large database from primary care networks with electronic health records for 17,278,392 patients representing nearly 40% of England’s population. This type of record typically includes information that may not be readily available from hospital or death records, including information on demographics, underlying medical conditions, smoking, weight and height.. The EHR records were linked to 10,926 COVID-19 deaths after deidentification. The researchers then performed statistical analysis to determine a hazard ratio (HR) for death from COVID-19 based on demographic, medical and social factors. One important aspect about this study that is different from many others is that it reports factors related to death from COVID-19 in the general population, not just those who test positive for the infection.
-
Advanced age was strongly associated with death from COVID-19; the hazard ratio (HR) for subjects over 80 years old was 20-fold higher than those aged 50-59. Men had a higher HR for death than women. Any non-white ethnicity had a higher HR for death from COVID-19 in both unadjusted and fully adjusted models. Several underlying medical conditions were notably associated with a higher HR for death from COVID-19, including diabetes, obesity, chronic heart disease, chronic respiratory disease and conditions leading to an immunocompromised state such as cancer, organ transplant, lupus and rheumatoid arthritis. A British social index score measuring deprivation, which combines scores related to employment, crime, education, poverty and income, was also strongly associated with death from COVID-19. A higher deprivation index score was associated with a higher HR for death.
-
The study names some of its own limitations. It combines lab-confirmed and probable COVID-19 deaths together due to lack of testing in some settings, which could lead to the inclusion of people dying of other causes in the analysis. Although the study is large and includes about 40% of England’s population, it may not be representative due to regional variation in choice of EHR used. The EHR records also had missing data, particularly for ethnicity, smoking and obesity.
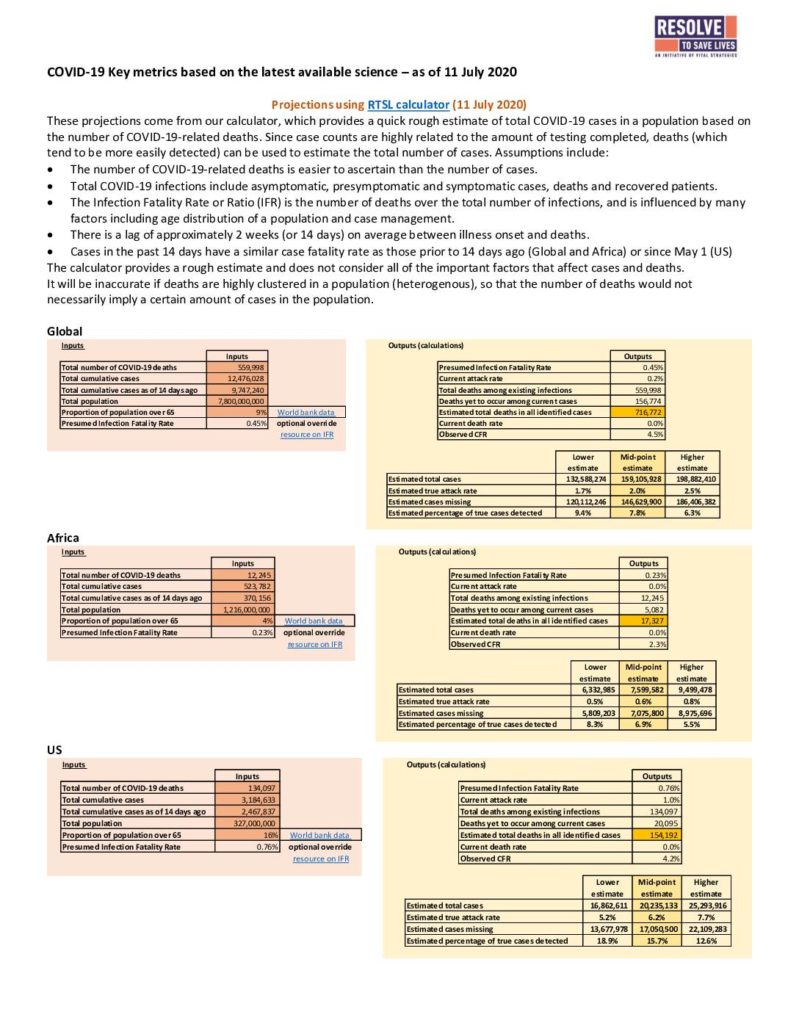
Global Assessment of the Relationship Between Government Response Measures and COVID-19 Deaths
(MedRxIV, preprint, July 4)
-
Researchers compiled publicly available data on daily COVID-19 case numbers and deaths as well as government responses in 170 different countries from January 1 to May 29, 2020. They developed a standardized composite stringency index to compare the extent to which nine different public health and social measures were adopted. Most countries eventually reached a composite index score of at least 80 out of 100. The composite index score was able to capture most of the difference in epidemic control measures implemented between countries. The speed of government response was measured by the number of days between the first recorded case of COVID-19 in a given country and when it achieved a composite index score of 40 out of 100.
-
Based on analyses conducted both within countries over time and across all 170 countries, the speed of response and the strength of restrictions, as measured by the composite index score, were strongly associated with slower daily epidemic growth rates. This association was robust even when investigators statistically controlled for key differences between countries such as demographic, economic, and health systems features. Over time, large numbers of COVID-19 cases and deaths that might have been prevented by more promptly introducing stringent public health and social measures, accumulated in many countries. For example, a one week delay in enacting policy measures to reach a composite index score of 40 may have led to 1.7 times as many deaths overall.
-
This analysis provides the most comprehensive multi-country evidence for the impact of public health and social measures in averting COVID-19-related mortality to date. Even so, the data are derived from observational studies and it is not possible to directly attribute causation. In addition, because so many countries implemented many measures at roughly the same time, it is not possible to isolate the contributions of individual interventions. More data on the role of specific measures may become available as countries relax restrictions incrementally.
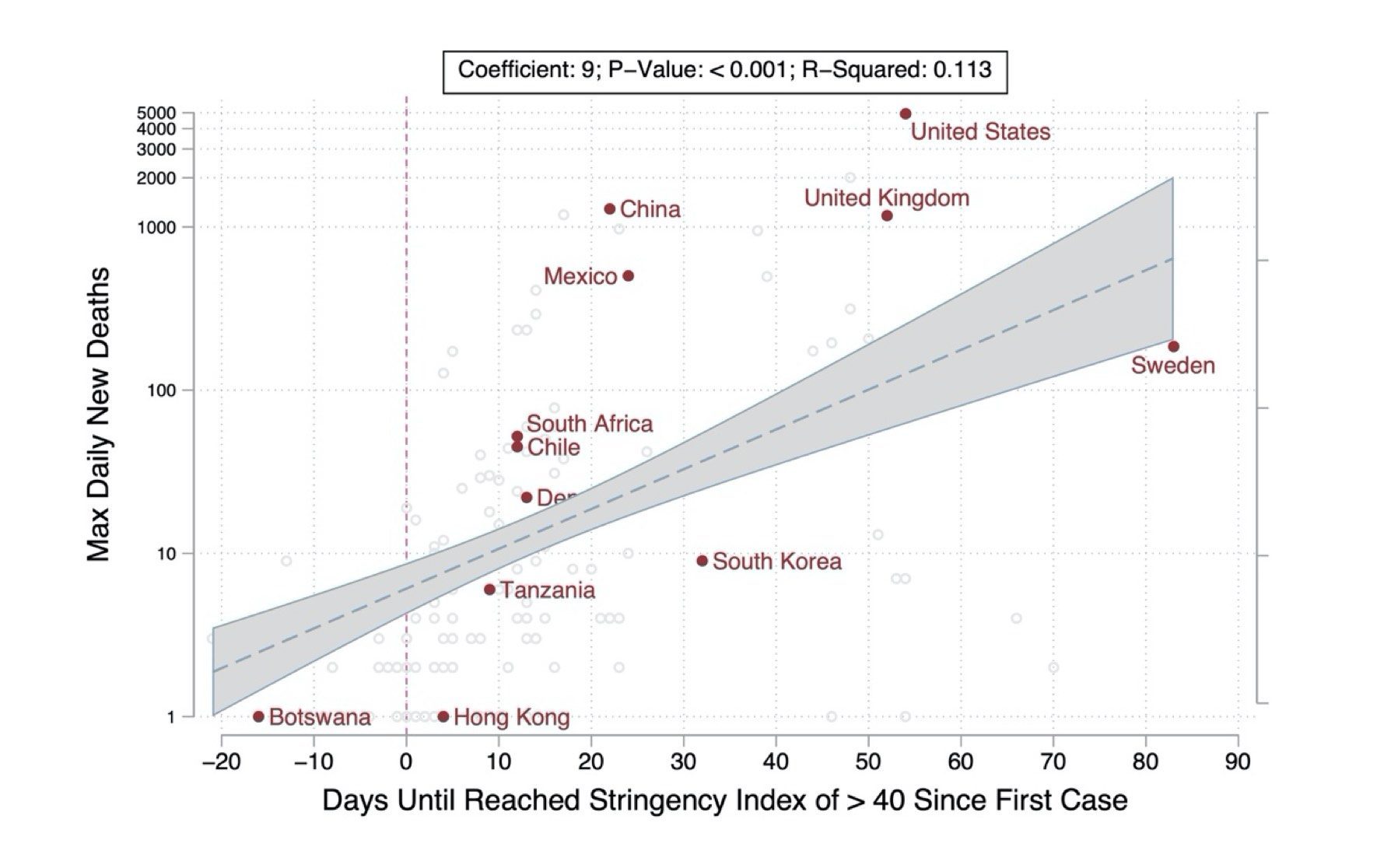
Maximum daily number of new COVID-19 deaths reported (at the peak of the epidemic) and delay between recording the first case and achieving a composite stringency index score of 40 out of 100 for nine public health and social measures in 170 countries, January through May 2020.
Prevalence of SARS-CoV-2 in Spain (ENE-COVID): a Nationwide, Population-Based Seroepidemiological Study
-
From April 27 to May 11, health authorities in Spain recruited 61,075 people from 35,883 randomly contacted households to participate in the national seroprevalence survey. Participants completed a short questionnaire about risk factors and symptoms of COVID-19, and took a point-of-care antibody test in their own homes. Some participants also submitted a blood sample for laboratory antibody testing.
-
Depending on the type of test used, between 4.6 – 5.0% of participants showed evidence of antibodies, or seropositivity, to SARS-CoV-2, indicating prior infection. Seropositivity was more common in urban areas such as in Madrid (>10%) compared to coastal areas (<3%). Children under 10 years old were less likely to be seropositive compared to other age groups. Around one third of those testing positive for antibodies did not report experiencing any symptoms thought to be associated with COVID-19. The majority (95%) of the Spanish population has likely not yet been infected despite the large numbers of COVID-19 cases. Seroprevalence was higher among those who had a confirmed COVID-19 case in their household.
-
This study is subject to misclassification, specifically with regard to analysis of self-reported symptoms, since a minority of patients with antibodies reported having a polymerase chain reaction (PCR) test. This study did not evaluate for protective immunity from infection. Of note, despite differences between the two testing approaches (point-of-care vs laboratory), the cheaper and faster point-of-care test yielded similar epidemiologic results at the population level.
Characteristics of Persons Who Died with COVID-19 — United States, February 12–May 18, 2020
(MMWR, Early release, July 10)
-
Given that death records do not always provide complete information about underlying medical conditions, clinical course, place of death and race and ethnicity, researchers at the U.S. CDC reached out to state and local health departments to obtain supplemental information about COVID-19 deaths that occurred between February 12 and May 18, 2020. They received information on 10,674 deaths from 16 jurisdictions across the U.S.
-
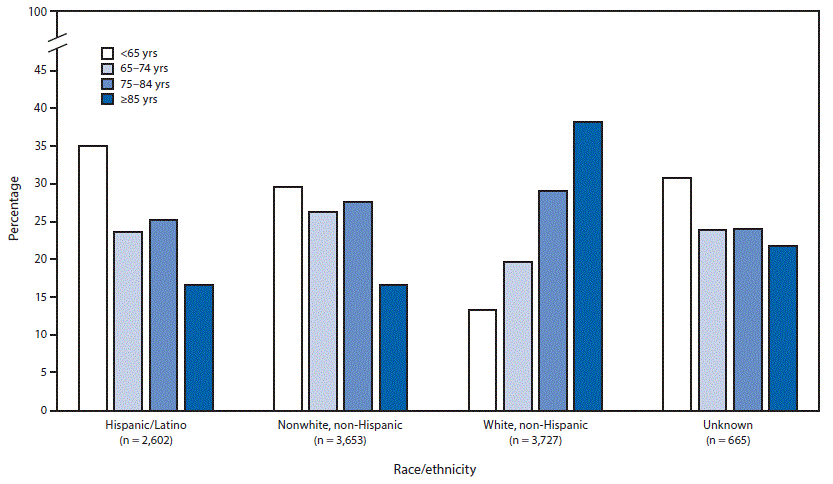
Men accounted for 61% of the deaths, and almost 80% of the overall deaths occurred in people 65 years old or greater. Age among decedents varied by race and ethnicity, with Hispanics and non-whites having a lower median age of death from COVID-19 than whites. The proportion of deaths at home or in the emergency department by age group was highest among people under 65 years old, while the proportion of deaths in a long-term care facility was highest among people 85 years old or greater. Most deaths occurred in persons with at least one (76%) or at least two (54%) underlying medical conditions. The most common underlying medical conditions were cardiovascular disease, diabetes, chronic kidney disease and chronic lung disease.
-
Despite obtaining additional supplemental data for this analysis, there were still missing data that limited further analysis and conclusions, especially for clinical course and underlying medical conditions. Reporting practices for some of these variables also vary by state. Mortality rates could not be calculated from this report. Changing patterns in the pandemic, including age of affected persons and impact of emerging therapies may be apparent in this type of data in the future.
CDC MMWR
The Implications of Silent Transmission for the Control of COVID-19 Outbreaks
(Proc Natl Acad Sci USA, July 6)
-
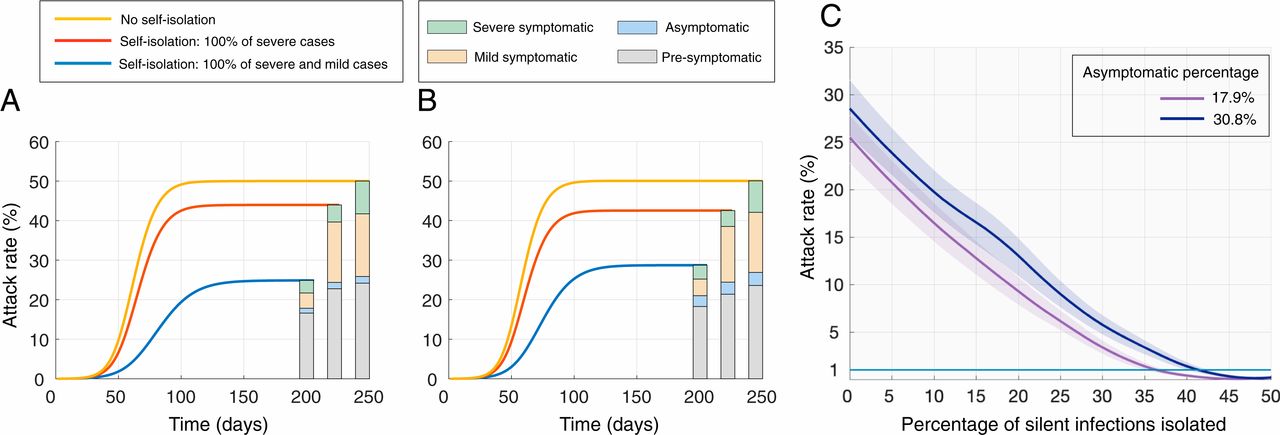
Recently published studies have established that asymptomatic people accounted for as much as 17.9% to 30.8% of all infections in well-characterized COVID-19 outbreaks. Researchers in this study used this range to simulate the proportion of transmission events that occur from asymptomatic, presymptomatic and symptomatic infections, including both mild and severe illnesses, in a population-based model. In each simulation, silent infections appeared to drive transmission, accounting for just over 50% of the overall attack rate. Presymptomatic infections contributed most while completely asymptomatic infections accounted for less than 10% of the overall attack rate.
-
The team also modeled the potential impact of identifying and isolating infected people and demonstrated how important silent infections can be for controlling transmission. Successfully identifying and immediately isolating every symptomatic case patient still resulted in an overall attack rate of 25% or more. It was possible to suppress the attack rate below 1% only when 33% to 42% of silent infections were also identified and isolated.
-
Although based on well-documented parameters, these estimates are drawn from simulated outbreaks; real-world experiences may require even higher levels of detection and isolation to suppress transmission. Even so, the study underscores the importance of case detection and contact tracing to detect and isolate infected people, even when they are otherwise healthy.
Increases in Health-Related Workplace Absenteeism Among Workers in Essential Critical Infrastructure Occupations During the COVID-19 Pandemic — United States, March–April 2020
(MMWR, July 10)
-
Researchers used data from a monthly national survey of 54,000 households conducted by the U.S. Census Bureau for the Bureau of Labor Statistics, to look for changes in expected versus observed health-related workforce absenteeism.
-
Overall, absenteeism in March and April 2020, a time when many parts of the US were being significantly affected by the COVID-19 pandemic, was not statistically different from the expected levels based on five-year baselines. However, in some occupations that are considered part of the nation’s essential workforce, absenteeism rose above expected and epidemic levels. This was most notable for health care support occupations, personal care and service occupations, and production occupations, which includes meat, poultry and fish processing workers. Absenteeism increased in occupations that are less able to avoid potential exposure to SARS-CoV-2 while largely remaining flat in other occupations.
-
The association seen in this study is not causal, but rather shows a temporal relationship. Health-related absenteeism may also be related to injury or non-COVID-19 illness. The analysis also does not take into account factors such as age, sex or race. Nevertheless, the data is correlated with that from other studies that have found increased risk from COVID-19 among health care personnel and food production workers.
Suggested citation: Cash-Goldwasser S, Kardooni S, Cobb L, Bochner A, Bradford E and Shahpar C. In-Depth COVID-19 Science Review July 4 – 10, 2020. Resolve to Save Lives. 2020 July 15. Available from https://preventepidemics.org/covid19/science/review/