Data insight
Past, current and future COVID-19 infections: the size of the iceberg and what tests can and can’t tell us
The public health response to COVID-19 is informed by available case and death counts and the modeled projections that use such data. Widely publicized projections of the global impact of COVID-19 were initially based on data from China available at the time. Predictions of the size of an epidemic change as the disease is better understood, surveillance methods improve, and testing increases. For example, the infection fatality rate (IFR), which conveys how likely an infected person is to die, tends to be revised downward over time as more people are tested, including those with asymptomatic or mildly symptomatic infections and those who were missed by early testing protocols. There are examples of this from the H1N1 and MERS epidemics. As COVID-19 epidemic mitigation efforts continue, it is critical to understand how many people have had COVID-19 infection and thereby generate accurate measures of mortality. The process of gathering these data at the population level entails widespread testing for COVID-19. The expansion of testing raises questions about the potential utility of determining each individual’s COVID-19 status and what that data can actually tell us.
| Setting | Currently infected (PCR) |
|---|---|
| Diamond Princess cruise ship | 19.2% |
| NYC pregnant women | 15.4% |
| USS Theodore Roosevelt ship | 14.7% |
| Vo, Italy | 4.4% |
| Iceland (general population) | 0.8% |
Two types of tests may establish whether someone has been infected with SARS-CoV-2. One type of test uses polymerase chain reaction (PCR) technology to detect the genetic material of SARS-CoV-2 and establish whether the virus is present. Most PCR testing has been performed on high-risk or symptomatic people to diagnose acute cases of COVID-19. If these data are used to estimate total COVID-19 case counts and fatality rates, adjustment to account for potentially undetected cases, including in people who were asymptomatic or had mild illness, is necessary. To avoid the uncertainty of these factors, one strategy is to do universal testing for SARS-CoV-2 in a given population, as was done among people on board the Diamond Princess cruise ship (19% tested positive) and the USS Theodore Roosevelt (15% tested positive). However, it is problematic to draw conclusions about community spread using data from those unique environments. Results of universal PCR testing among pregnant women in New York City suggested that the community prevalence of acute COVID-19 infection, at least among people for whom this population was representative, may have been about 15%. In Iceland, where 6% of the total population was tested through a combination of targeting high-risk individuals and screening the general population, 1,221 (13.3%) of the 9,199 targeted high-risk individuals tested positive, and 100 (0.8%) of the 13,080 members of the general population tested positive. In Italy, PCR testing of the majority of one town’s population suggested that 4.4% (95% CI 3.6-5.3%) of people were infected and had the virus detectible by PCR. These studies have also provided important data on the proportion of infections that are asymptomatic upon detection (47% of infected people on the Diamond Princess Cruise, 43% of Icelanders who tested positive during general population screening, 89% of infected pregnant women in New York City, 60% of infected people on the USS Theodore). Of note, distinguishing infections detected during a pre-symptomatic phase from those that are truly asymptomatic requires follow up over the duration of illness. Modeling approaches can also be used; for instance, it was estimated that the asymptomatic proportion of cases on the Diamond Princess was 18%.
| Setting | Ever infected (Serology) |
|---|---|
| Chelsea, MA, USA | 32.0% |
| New York City, NY, USA | 21.2% |
| Gangelt, Germany | 14.0% |
| New York State, USA | 13.9% |
| Los Angeles, CA, USA | 4.1% |
| Santa Clara, CA, USA | 2.8% |
The second type of test for infection with SARS-CoV-2, a serology test, detects antibodies that are produced in response to infection. PCR tests detect whether virus is currently present; serology tests detect infections that have either been present for a few days or already resolved. Following the development of SARS-CoV-2 antibody tests, studies on the proportion of the population with antibodies (seroprevalence surveys) have generally suggested that official case counts underestimate infection prevalence. Seroprevalence data from Santa Clara County, California, led investigators to estimate a cumulative total of 48,000 – 81,000 infections in the county, which is 50 – 85 times higher than the official COVID-19 case count (~1,000 cases), as of April 1. Seroprevalence data from Los Angeles County, California, suggests a cumulative total of 220,000 – 442,000 people were infected (rather than the official count of 8,000 cases) by early April. In Chelsea, Massachusetts, 32% of 200 people recruited on the street had antibodies to SARS-CoV-2. In Gangelt, Germany, 14% of the population had antibodies. In New York State, 14% of state residents and 21% of New York City residents who underwent serologic testing were positive for SARS-CoV-2 antibodies, suggesting that as many as 2.7 million state residents may have been infected (rather than the official count of ~270,000). Some of these prevalence estimates were used to adjust the IFR, yielding results (Santa Clara County: 0.1 – 0.2%; Los Angeles County: 0.1 – 0.3%; Gangelt, Germany: 0.37%) lower than prior estimates. As with the results of PCR-based studies, caution must be utilized when extrapolating seroprevalence results to a larger population. For example, in the Santa Clara County study, participants were recruited on Facebook, which skewed the sample by race, sex, and zip code when compared with the county population. In Germany and Massachusetts, the towns in which studies were conducted have been particularly hard-hit by the epidemic. The New York study recruited participants at locations such as supermarkets, so these individuals may have had a higher risk of infection than others. Many seroprevalence studies are planned or underway, including a Centers for Disease Control and Prevention (CDC) study, a National Institutes of Health (NIH) study, studies in the Netherlands and Switzerland, and a multi-country study conducted by the World Health Organization (WHO).
The possibility of identifying those who were infected in the past raises questions about other potential uses of SARS-CoV-2 antibody tests. A number of SARS-CoV-2 antibody tests are available for use or in development. There are laboratory-based serologic tests that can quantify antibodies or even detect antibodies that are able to neutralize virus. There are also serologic tests designed as rapid diagnostic tests (RDTs), similar to home pregnancy tests. These tests can garner a lot of attention given their low cost, ease of use, no requirement for a laboratory, and quick result turn-around time. The design of an RDT to detect antibodies is pictured below.
One potential use of antibody tests is to more accurately diagnose COVID-19 patients. False negative PCR results may occur, particularly later during the course of infection, which is reflected in CDC guidelines that infected healthcare workers should ideally have two consecutively negative PCR test results before returning to work. That said, serology tests have their own time window of reliability and are generally unable to detect early infection; one summary of available evidence suggests that the median time between COVID-19 symptom onset and detection of antibodies is 11 days (IQR 7.25-14 days). in most COVID-19 patients in several studies, antibodies were not reliable detected until approximately 7 days after the onset of symptoms. Antibody-based tests may be used to assess vaccine efficacy and to screen convalescent COVID-19 donor plasma for antibodies; the Food and Drug Administration (FDA) currently recommends that plasma donation may proceed if testing for neutralizing antibodies is not available. The presence of antibodies may have implications for healthcare and other frontline workers, and ‘immunity certificates’ have been discussed in several countries. However, it is not known if the presence of SARS-CoV-2 antibodies signifies immunologic protection, as there are components of the immune system other than antibodies. For example, in a study of 175 patients who recovered from mild COVID-19, about 30% generated very low titers of protective antibodies known as neutralizing antibodies, and in 10 (5.7%) patients, neutralizing antibodies were undetectable. It is also unclear whether infection with SARS-CoV-2 protects from re-infection. There are limited data from macaques that prior COVID-19 protects from re-infection; what observational data on COVID-19 reinfection in humans suggests isn’t clear. There were no comprehensive observational studies of SARS or MERS re-infection in humans because each epidemic had a single peak. There is evidence that re-infection with endemic coronaviruses (viruses that can cause the common cold) can occur though with reduced or absent symptoms. However, definitive conclusions about COVID-19 cannot be drawn from these data. Clearly, further studies on the nature, extent, duration, and protection of immune responses to SARS-CoV-2 are needed. For these reasons, WHO released a science brief advising against immunity certificates, since there is currently no evidence that people who have recovered from COVID-19 and have antibodies are protected from a second infection.
The FDA is utilizing an emergency use authorization process and has relaxed guidelines around the use of yet-to-be evaluated serologic tests, as long as the test developer evaluates the performance of the test and labels it as not intended as the sole basis for diagnosis. Generally, the performance of a new diagnostic test must be compared with a test considered accurate (the ‘gold standard’) by performing both tests in a sample of patients with and without the disease. Comparing these results can determine the ability of a test to correctly identify those who have the disease (the ‘sensitivity’ of the test) and to correctly identify those who do not have the disease (the ‘specificity’ of the test). This was done in the Santa Clara County seroprevalence survey: a new commercial RDT was utilized, so investigators generated and collected data on the performance of the RDT (pooled sensitivity ~80%; pooled specificity ~99%) and used those data to adjust their final results. A study on the performance of nine commercial SARS-CoV-2 antibody assays found that the lab-based tests generally performed well, but many RDTs did not. Similarly a review of 12 serologic tests found variable performance affected by subjective interpretation of the test bands. Indeed, some serologic tests purchased by governments and companies have not delivered as promised in terms of diagnostic accuracy. The FDA, CDC, and NIH are collaborating to establish capability to evaluate tests for manufacturers. For now, the reliability of most serology tests on the market is uncertain. The WHO has published recommendations against using SARS-CoV-2 antibody RDTs for patient care, and other public health organizations have cautioned against reliance on the results of antibody-based tests alone.
In-depth topics
COVID-19 testing prioritization in the United States
Robust testing is needed to track and respond to the COVID-19 pandemic. Unfortunately, many locations have testing capacity constraints due to supply shortages, testing backlogs, test quality issues, and logistical challenges. Until there is much more testing capacity, jurisdictions and health care providers should prioritize people to be tested based on health benefit. Testing is highest priority if it may improve clinical outcomes or contribute to reduced spread of disease (particularly to medically vulnerable people). Additional testing will require health care and public health system capacity to follow up on positive results. The size of each group and therefore the number of tests needed, as well as the number of tests available, are also important considerations. Initially, only the highest priority groups should be tested. As testing becomes more widely available, testing should be expanded to other groups in a stepwise fashion, based on their priority.
COVID-19 testing is an essential component of the Box It In strategy to support the rapid isolation of cases, elicitation of contacts, and quarantine of contacts. We prioritized testing groups and identified how many tests would need to get done per group. For the United States, the groups in Priority I represent approximately 350,000 – 700,000 tests per day (more if there are more cases and contacts), Priority II an additional 2.8 million, Priority III an additional 1 million, and Priority 4 an additional 6.5 million. As testing capacity is being expanded, groups for testing should be prioritized as outlined in the brief in order to maximize the goals of saving lives and preventing spread of infection.
For more information see our brief on testing in the United States
Tracking the virus, its genome, and mutations - a critical aspect of disease surveillance.
The novel coronavirus SARS-CoV-2 belongs to a family of RNA viruses that can cause a range of disease in humans and animals. Common human coronaviruses cause mild disease and circulate in many parts of the world. Recently, novel coronaviruses, likely zoonotic in nature (of animal origin), have changed in such a way to cause disease in humans which can be severe, as with SARS-CoV-1 in 2003, MERS-CoV in 2012 and thereafter, and now SARS-CoV-2. SARS-CoV-2 is a relatively large RNA virus and has about 29,800 – 30,000 base pairs of genetic material arranged in a single linear segment coding for 27 proteins. This genetic material is the key for the molecular components that translate into the virus’ structure and function, including its ability to bind to and enter human cells and cause disease.
All viruses mutate as part of their life cycles. These mutations have been compared to typographical errors that are not picked up and corrected. RNA viruses such as those that cause COVID-19, measles and the flu mutate more often than DNA viruses such as those that cause chickenpox, hepatitis B, or herpes.
Mutation. The word naturally conjures fears of unexpected and freakish changes. Ill-informed discussions of mutations thrive during virus outbreaks, including the ongoing spread of SARS-CoV-2. In reality, mutations are a natural part of the virus life cycle and rarely impact outbreaks dramatically.Nathan Grubagh – Yale School of Public Health
Although some researchers can make conjectures about the meaning of specific detected mutations, these are speculative in nature. They are not based on observing how the virus is behaving in humans and populations over time, but rather on how the virus may behave in a controlled lab environment in one snapshot. Experts agree that although there is always a chance for a mutation to result in a change in the virus’ behavior, this type of activity has not been documented yet for SARS-CoV-2 – and it is an important reason why some groups that collect and analyze open source data about the genetics of the virus monitor these mutations closely.
Significant genetic mutations could have major implications for vaccine development and prevention as well as for therapeutic targets and treatment. Currently, thousands of sequences of virus genetic code from COVID-19 patients around the world are being sent to organizations collecting, compiling, and analyzing viral genomics. Such efforts are being led by Nextstrain and GISAID, offering near real-time information about the evolution of the outbreak. In addition to being able to monitor for potentially serious mutations, this type of surveillance also allows scientists to track the virus and its movements, how it is spreading, and contribute to improving outbreak response.
The bottom line for now is that we must continue to monitor this additional data, identify changes in the genes of the virus, and look for clues into the effect these changes may have on virus spread and behavior and implications for outbreak response. It is of crucial importance to correlate genetic changes in the virus with different epidemiologic patterns of the disease in humans.
COVID-19 and Noncommunicable Diseases
Data from China, Italy and the US indicate that patients with noncommunicable diseases (NCDs) or associated conditions make up the large majority of patients who have been either hospitalized or who have died from COVID-19. Further, systematic reviews have indicated that noncommunicable diseases (NCDs) such as hypertension, chronic obstructive pulmonary disease, cardiovascular disease, and diabetes are associated with increased risk of either severe disease or death. Age is the strongest predictor of COVID-19 mortality; age is also strongly associated with NCD prevalence, and most studies to date have failed to take age into account when looking at the impact of NCDs. The few studies that have controlled for age have, for the most part, continued to find a relationship with at least one NCD and COVID-19 severity or mortality. The largest, a study of more than 4,000 patients with positive COVID-19 tests in a single US hospital system, found that obesity, diabetes, and kidney disease were significant predictors of hospitalization and obesity and diabetes significant predictors of death among those hospitalized. Overall, however, while existing data is suggestive, it remains unclear which NCDs are implicated as a cause of severe COVID-19
For more information on NCDs and COVID-19, see our supplement
Other infectious diseases and COVID-19
To date there are few studies that look at whether diseases such as tuberculosis, malaria, or HIV have any impact on the risk of COVID-19 disease severity or mortality. There have been a few case reports describing individual patients co-infected with HIV and COVID-19, but so far the total number of patients included in these reports is only eight total: three in China (Shenzhen, Wuhan) and five in Spain. Six of the patients were on combination antiretroviral therapy (ART) before COVID-19, two with protease inhibitors (darunavir/cobicistat); two were ART-naïve, both with a low CD4 count. Three of the cases (including both ART naïve cases) were severe and required oxygen (one intubated), but none were reported to have died. During their illness, seven of the eight patients were put on an ART regimen containing the protease inhibitor lopinavir-ritonavir, which is being investigated as a SARS-CoV-2 antiviral medication. One of the patients consistently tested negative for SARS-CoV-2, but later tested positive for antibodies. Although some authors hypothesize that people living with HIV may be more susceptible to COVID-19, others have hypothesized that immunosuppression may actually protect from the severest forms of disease. To date, there is no evidence on COVID-19 and HIV from Africa, despite the high prevalence of HIV in many African countries. A meta-analysis looking at other, non-COVID-19 acute respiratory tract infections (including other coronaviruses) found that case fatality rates (CFR) were significantly higher among people living with HIV, particularly in children under 5 (OR 5.5 compared to OR 1.5 for older children and adults).
FAQs
Can I wear contact lenses during the COVID-19 pandemic?
Despite news reports suggesting that all contact lens wearers should switch to glasses to prevent the spread of SARS-COV-2, according to the CDC, the American Optometric Association, and the American Academy of Ophthalmology, there is no evidence that wearing contact lenses increases the risk of COVID-19. Contact lens wearers should wash their hands for 20 seconds with soap and water before putting on or taking off lenses. They should also follow recommended contact lens hygiene practices. According to US CDC, hydrogen peroxide-based systems for cleaning, disinfecting, and storing contact lenses should be effective against the virus that causes COVID-19. For other disinfection methods, such as multipurpose solution and ultrasonic cleaners, there is currently not enough scientific evidence to determine efficacy against the virus. Some contact lens wearers touch their face more frequently when wearing contacts; in this case, it may be a good idea to wear glasses or otherwise minimize touching eyes with unclean hands.
Article summaries
Epidemiology
Suppression of COVID-19 outbreak in the municipality of Vo’, Italy
(MedRxiv preprint, 17 April 2020)
- In Vo’, Italy (total population 3,275), the point prevalence of SARS-CoV-2 infection diagnosed by PCR was 2.6% (73 positive of 2,812 tested) and 1.2% (29 positive of 2,343 tested) at the respective beginning and end of a 2-week lockdown period. Models suggested that at least 4.4% (95% CI 3.6-5.3%) of the population of Vo’ had been infected with SARS-CoV-2 by the end of the study.
- Eight of the 29 positive results in the second survey identified new cases. Transmission chain reconstruction suggested that the majority of those were infected in the community before the lockdown or from asymptomatic cases living in the same household.
- Over 40% of cases were asymptomatic. The presence of symptoms was not associated with viral load or with time to viral clearance (average time 9 days from symptom onset).
- The authors calculated an initial weekly effective reproduction number of 3.0 (95% CI 2.5-3.5) which declined to 0.14 (95% CI 0.0-0.29) by the end of the lockdown.
COVID-19 Antibody Seroprevalence in Santa Clara County, California
(MedRxiv preprint, 17 April 2020)
- Of 3,330 healthy community dwellers in Santa Clara County recruited over 24 hours on April 3-4, 2020, 50 (1.5%) tested positive for antibodies to SARS-CoV-2.
- Investigators adjusted results to account for the following:
- Population characteristics: Facebook advertisements for recruitment were targeted by zip code and sociodemographics, but the study population ultimately differed significantly from the County population by race, sex, and zip code. Prevalence estimates were adjusted to account for this.
- Assay performance: Investigators used a commercially produced lateral flow assay (LFA) to test for antibodies to SARS-CoV-2 and adjusted results based on how well the tests performed as assessed by: 1) the investigators, 2) the manufacturer, and 3) a combination of both (combined sensitivity was 80.3% (95 CI 72.1-87.0%) and combined specificity was 99.5% (95 CI 98.3-99.9%)).
- After adjusting for population characteristics and test performance, the prevalence of SARS-CoV-2 antibodies was 2.75% (95% CI 2.0%-3.5%). The authors concluded that while the reported number of confirmed COVID-19 cases in Santa Clara County on April 1 was 956, their data suggest that the true number of infections was 50-85 times higher (for a total of 48,000-81,000 infections) and that the infection fatality rate may be much lower (0.12 – 0.2%) than reported case counts would suggest.
COVID-19 Outbreak associated with air conditioning in restaurant, Guangzhou, China, 2020
(EID, 2 April 2020)
- The figure below shows the seating of case patients and other family members on January 24, 2020.
- Families A and B were both present in the restaurant for an overlapping interval of 53 minutes; Families A and C for 73 minutes.
- Neither Family B nor Family C had any other history of exposure, and community transmission was not documented in Guangzhou at this time.
- It is likely that at least one member from each Family B and Family C was infected at the restaurant (infections among other family members may have occurred from later contact with one another), suggesting that infectious virus was propelled farther than 1m (3 ft).
- Airborne transmission (via aerosols) is less likely: 8 restaurant staff and another 73 customers present in the restaurant at the same time were not infected. Viral RNA was not detected in samples taken from the air-conditioning system.
Figure: Sketch showing arrangement of restaurant tables and air conditioning airflow at site of outbreak on January 24. Red circles indicate seating of eventual case-patients, yellow-filled red circle indicates index patient.
Coronavirus Disease Outbreak in Call Center, South Korea
(EID, August 2020)
- The article describes an epidemiologic investigation of a cluster of cases at a call center in a 19-story mixed-use building in Seoul, Korea.
- Of 1,145 persons they investigated, 97 (8.5%) were confirmed to have COVID-19
- The attack rate on the 11th floor where 97% of cases were found was 43.5%
- The household attack rate among contacts of case-patients (average 2.3 household contacts per case) was 16.2%
Figure: Floor plan of the 11th floor of the call center. Blue colored seats indicate confirmed cases.
COVID-19 Outbreak Among Three Affiliated Homeless Service Sites – King County, Washington, 2020
(MMWR Early Release 22 April)
- Once a shelter resident tested positive after presenting to a local hospital with acute encephalopathy and cough, fever, and shortness of breath, testing was offered to all other staff and residents in the shelter network, which consisted of three shelters that shared activity space during the daytime.
- Two rounds of testing and screening were performed at the shelters. A small number of persons were also tested outside the shelter system. In total, 195 residents and 38 staff were assessed, identifying 35 confirmed or probable cases among residents and 8 among staff.
- A high proportion of cases among shelter staff and residents were identified through special testing events planned and implemented by local and federal public health, in conjunction with shelter leadership and management. This allowed for prompt identification of additional cases and appropriate isolation.
- Although the surrounding community was under stay-at-home orders, physical distancing was more difficult to implement in the shelter setting due to a lack of bathing and hygiene facilities at the overnight-only shelter. One shelter was closed. Then, there was an assessment of the available space, staff, and facilities which allowed for appropriate isolation of symptomatic persons and positive cases and access to basic needs in the remaining facilities.
The characteristics of household transmission of COVID-19.
(CID. 17 April)
- Through medical record review, serial SARS-CoV-2 RT-PCR testing, and telephone interviews of 105 index cases and their 392 household contacts, the authors identified 64 SARS-CoV-2 positive household contacts making for a secondary attack rate of 16.3%. Nine of these contacts (14.1%) were asymptomatic despite testing positive.
- Children living in the same household as an index case were less likely than adults to become infected (4% for under 18 yrs vs 17.1% for >=18 yrs contacts, OR 0.18; 95% CI 0.08 – 0.54). Among adult contacts, spouses were more likely than non-spouses to become infected (27.8% vs 17.3%, OR 2.27; 95%CI 1.22-4.22).
- When the index case self-isolated immediately at the onset of any symptoms, as was done in 14 cases, there was no secondary transmission to other household members. These cases were reportedly isolated in the household with a mask, and separate eating and living quarters.
- Secondary transmission within the household for SARS-CoV-2 was higher than for SARS-CoV-1, MERS-CoV, and similar to that of 2009 H1N1 influenza (13%).
Assessment of SARS-CoV-2 Infection Prevalence in US Homeless Shelters – Four US Cities, March 27 – April 15 2020
(MMWR Early Release 22 April)
- RT-PCR testing of all residents and staff for SARS-CoV-2 was performed at participating shelters through coordination among local public health infrastructure, homeless services providers, academic partners and health care providers. In total, 1,192 residents and 313 staff were tested from 19 shelters.
- In Boston and San Francisco, testing was done at shelters already known to have a cluster of cases in the preceding 2 weeks. In Seattle, testing was performed at shelters known to have a cluster as well as those where only a single case had been identified. In Atlanta, testing was performed at shelters not known to have had any cases in the preceding 2 weeks.
- When testing was done after a cluster of cases had already been identified, the additional number of infected persons identified during testing was higher than when testing was done before the identification of a cluster.
- Early testing and implementation of control strategies can aid in controlling or preventing spread of disease in homeless shelters.
Presymptomatic SARS-CoV-2 Infections and Transmission in a Skilled Nursing Facility
(NEJM, 24 April 2020)
- Residents of one skilled nursing facility were repeatedly tested to assess disease transmission and the adequacy of symptom-based screening to identify residents who were infected
- Overall, 48 (63%) of 76 residents who were tested (PCR, viral culture, sequencing) were found to be positive of SARS CoV-2.
- Of these 27 (56%) were asymptomatic at time of testing, 24 (89%) of whom later developed symptoms (median onset 4 days later) and were considered presymptomatic.
- Median cycle threshold values for the four symptom status groups were similar (asymptomatic residents, 25.5; presymptomatic residents, 23.1; residents with atypical symptoms, 24.2; and residents with typical symptoms, 24.8) and virus was isolated from 17/24 presymptomatic and 1/3 asymptomatic residents.
- The doubling time among residents was estimated to be 3.4 (95% CI 2.5-5.3 days) days, in comparison to 5.5 (4.8-6.7) days in the surrounding county.
- Overall, the facility had a 26% case-fatality rate, and 19% of staff members were diagnosed with infection.
A systematic review of antibody mediated immunity to coronaviruses: antibody kinetics, correlates of protection, and association of antibody responses with severity of disease.
(MedRxiv Preprint, 17 April 2020)
- The median time to detection of antibodies for SARS-CoV-2 is 11 days (IQR 7.25-14), similar to SARS-CoV-1, but shorter than for MERS-CoV.
- Long-term studies in other coronaviruses have shown the persistence of antibodies, specifically IgG, past one year post-infection and up to 3 years. A longer duration of antibody persistence has been associated with more severe symptoms during illness.
- In other human coronaviruses, waning immunity has been shown, where it has been possible to induce a reinfection 1 year after initial infection. The reinfection was however associated with lower severity.
- The knowledge gap around seropositivity and its correlation to protection or immunity needs to be filled prior to use of policies such as immunity passports.
Clinical features
Symptom Screening at Illness Onset of Health Care Personnel With SARS-CoV-2 Infection in King County, Washington.
(JAMA, 17 April 2020)
- Interviews were conducted with 48 of 50 SARS-CoV-2 positive HCP in the King County, Washington area to assess symptoms at the time they first became ill compared to symptoms they experienced over the course of the entire illness. When asked to recall their symptoms the day they first felt ill, 50% reported cough, 41% reported fever, and 10.4% reported shortness-of-breath, compared to 87.5%, 75%, and 31.3% reporting these symptoms, respectively, over the course of the entire illness.
- Eight of the HCP interviewed (16.7%) did not have cough, fever, shortness-of-breath or sore throat on the first day of illness. Of these, 7 eventually developed one or more of these symptoms between 1-7 days after first feeling any symptom (median 2 days).
- Almost two-thirds (64.6%) of the HCP interviewed reported working at least 1 day while having any symptom (median 2 days, range 1-10 days).
- More inclusive criteria for screening and testing, especially for ill HCP, may result in better detection of those infected with SARS-CoV-2. Face mask use by all HCP may improve source control and reduce transmission from those who display atypical or minimal symptoms, or no symptoms at all.
Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area
(JAMA, 22 April 2020)
- From March 1 to April 4, 2020, 5700 patients with a lab-confirmed diagnosis of COVID-19 were admitted to a network of 12 hospitals in the New York City area. The average age of these patients was 63 years and 61% were male. The most common comorbidities were hypertension, obesity and diabetes.
- A subset of these patients whose disposition (death vs discharge) was already available was further analyzed for clinical outcomes. Among these 2,634 patients, 14% were treated in the ICU, with 12% receiving mechanical ventilation. Mortality for those requiring mechanical ventilation was 88%, and overall mortality was 21%. More than half of the patients in the study did not have disposition data available since they were still hospitalized, which could result in a lower overall mortality associated with mechanical ventilation.
- Among those receiving mechanical ventilation, mortality was higher for older patients – 97% for those 65 years or older, and 76.4% for those 18-64 years of age. There were no deaths among patients younger than 18 years of age. In addition, men had a higher overall mortality compared to women when stratified by age groups.
- The overall readmission rate among those discharged was 2.2%. Older patients were more likely to be readmitted.
Suggested citation: Cash-Goldwasser S, Kardooni S, Cobb L, Bochner A, Bradford E and Shahpar C. In-Depth COVID-19 Science Review April 18 – 24, 2020. Resolve to Save Lives. 2020 April 27. Available from https://preventepidemics.org/covid19/science/review/
Download this review
-
Science Review April 18-24 2020
(1.22 MB)
-
Science Metrics April 27 2020
(380.91 KB)
-
Supplement on the impact of NCDs on COVID-19
(190.24 KB)
-
COVID-19 Testing Prioritization in the United States
(90.06 KB)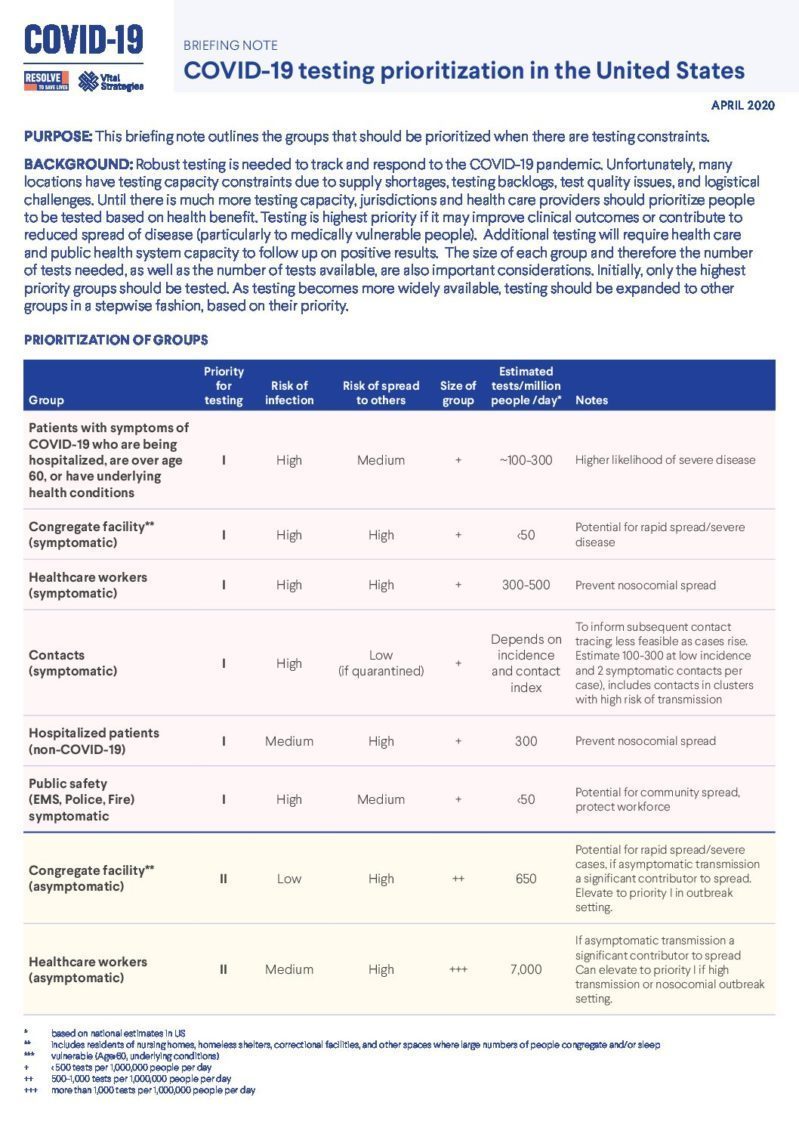
-
Science Review French Apr 18–24 2020
(725.25 KB)
-
Science Review Spanish April 18 - 24 2020
(765.25 KB)
-
Science Review Portuguese Apr 18 - 24 2020
(483.86 KB)







