In-depth
COVID-19 and mass transit
In many urban centers, people rely on mass transit to get to work, essential appointments, and other destinations. With COVID-19, fear of getting infected on mass transit, combined with the fact that many people continue to work from home, has led to major drops in ridership and has threatened the financial stability of many mass transit agencies. A recent report commissioned by the American Public Transit Association concluded that the risk of contracting COVID-19 on mass transit is minimal and that it “had little or no role in the spread of the disease.” Although these findings are encouraging, overall the evidence remains limited on COVID-19 transmission and mass transit. In this review, we summarize the current state of the evidence.
Mass transit as it is currently being used is NOT a major driver of COVID-19
The American Public Transit Association report explored whether there were correlations between mass transit ridership levels and COVID-19 cases in seven U.S. cities and metro areas. They did not conduct statistical tests, but the graphs of these metrics do not appear to show a relationship between the two. If public transit was driving the epidemic, one would expect to see curves that mirrored each other, with a one- to two-week lag between ridership and cases. The report also looked at cities such as Singapore, Hong Kong, Tokyo, Seoul and Paris that are densely populated and most people use public transit. In Singapore, Tokyo and Seoul, lower rates of COVID-19 made it easier for public health officials to identify clusters; none of these have been linked to the public transit systems. Similarly, in Paris, only five cases had been linked to transport through August (and these were not necessarily intracity mass transport).
A working paper originally released in April suggested that the New York City subway system was the major source of spread of the epidemic in New York City during the month of March by looking at the correlation between ridership declines by subway line and infection rates by zip code. Although suggestive, the paper was not able to distinguish between people who were exposed to COVID-19 on the subway versus at their destination. Further, the article covered a time before the mask mandate in New York City and therefore may not be relevant to current conditions.
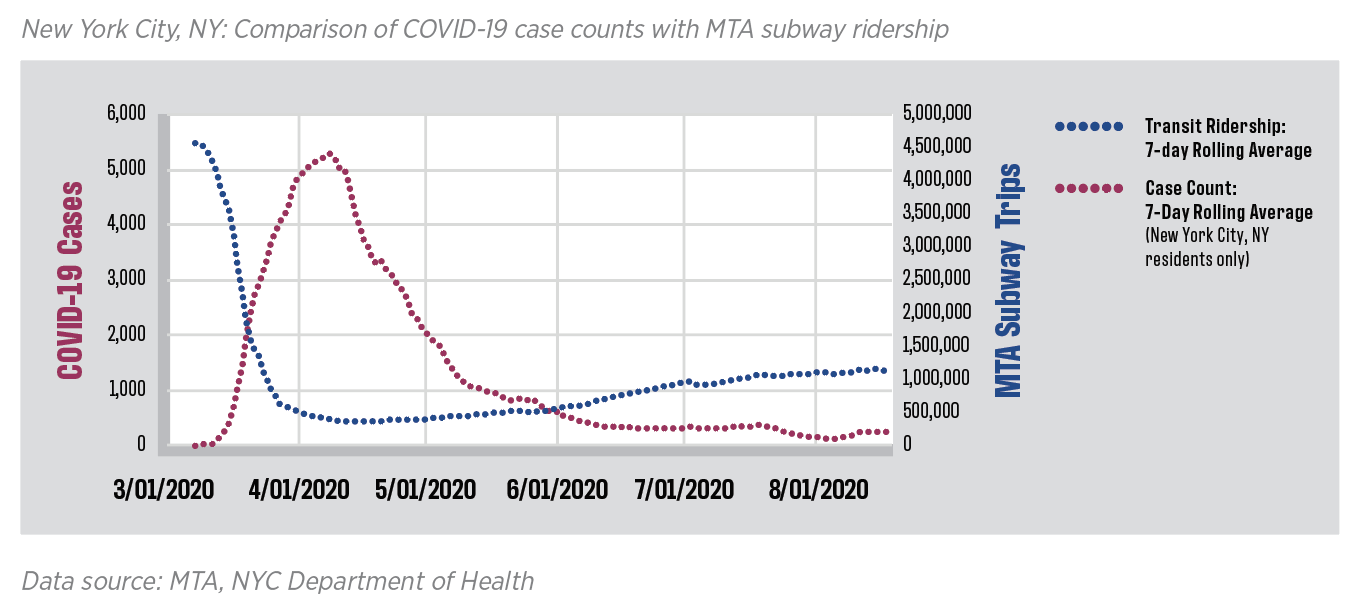
American Public Transportation Association and Sam Schwartz Consulting
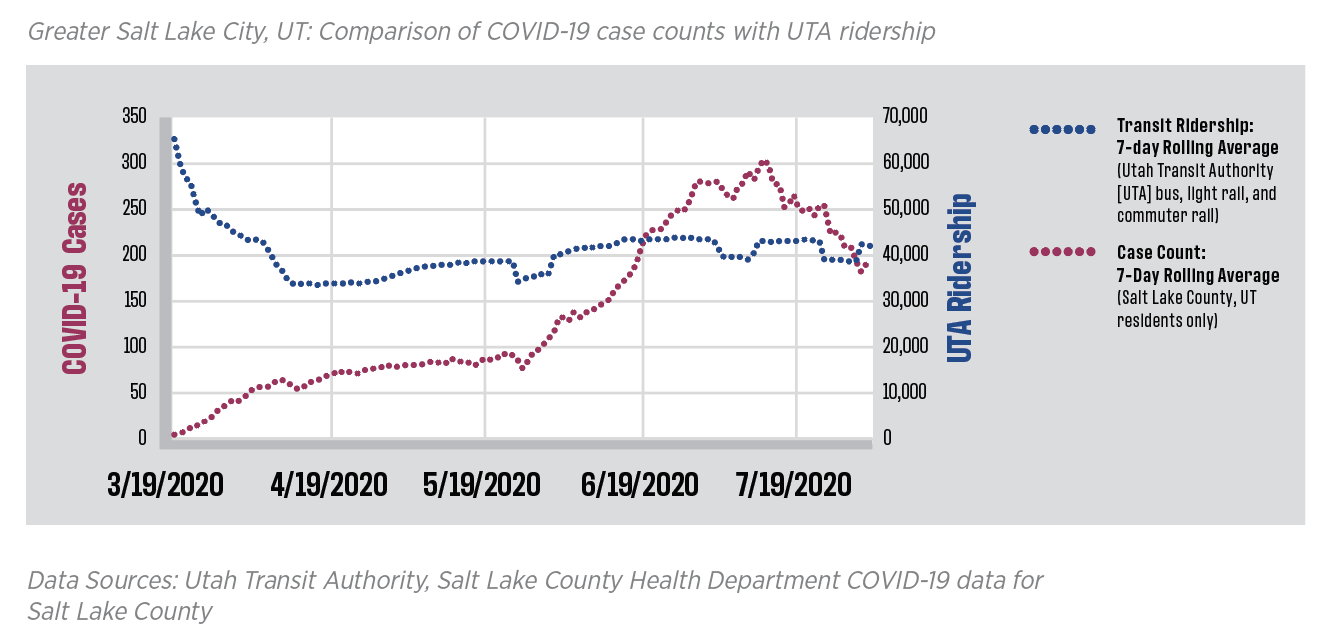
American Public Transportation Association and Sam Schwartz Consulting
Risks of riding on mass transit
The evidence presented in the American Public Transit Association report illustrates that mass transit is not a major driver of COVID-19 at this stage in the epidemic, but it does not mean that transmission is not occurring. Most contact tracing systems cannot identify passengers who might have ridden together on a bus or subway train, so it is unlikely that transmission occurring on mass transit will be identified.
As with any other setting, risk of transmission can be reduced by following the 3 W’s: wearing a mask, washing hands and watching distance from other people. Recent guidance from U.S. Centers for Disease Control and Prevention on how people can protect themselves while on public transit emphasizes these precautions. Mask compliance on many transit systems appears to be relatively high, but it can be difficult, if not impossible, to maintain at least 6 feet of distance between riders, particularly during rush hour.
Although physical distancing would be ideal, mass transit may still be relatively low risk at this time due to the following:
- Most people ride alone and do not speak. Although speaking quietly is not very different from breathing, speaking (or singing) loudly increases the production of infectious particles.
- Although buses and trains are enclosed spaces, ventilation is often better than in many indoor spaces, reducing the likelihood of spread in case the virus is present in aerosols. For instance, in New York City, subway cars replace their air completely 18 times per hour. Further, opening doors frequently can help ventilation.
- Many trips are short and exposure to any one individual is even shorter, unlike travel on long-distance trains, planes or buses. The duration of exposure to an infected person is a critical factor in whether a person gets infected.
- Ridership is down, allowing for more physical distancing while riding and reducing the likelihood that an infectious individual will ride. Although maintaining 6 feet of distance between riders is often not possible, ridership is nowhere near its peak. The American Public Transit Association case studies report ridership in major Asian cities ranging from 63% of normal levels in Tokyo in August to 76% in Hong Kong. In New York City, subway ridership is still only 30% of 2019 levels.
Finally, as with every other setting, the risks of contracting COVID-19 while riding mass transit will depend on the level of COVID-19 in the community. Particular care should be taken when community transmission is high.
Benefits of mass transit
Although the risks of getting COVID-19 while using mass transit may be higher than modes of transportation that don’t require interacting with other people, mass transit has other health benefits. On a per mile basis, car travel is 10 times more dangerous when it comes to injuries or deaths from crashes. Using public transit also increases physical activity and decreases air pollution, leading to health benefits such as reduced cardiovascular and respiratory diseases. Finally, for many who don’t have cars and/or who live in urban areas where car travel is impractical, it may be the most practical way to get to work or other necessary destinations.
*Resolve to Save Lives and the Partnership for Healthy Cities developed a guide is designed to help cities reduce the risk of COVID-19 transmission within urban mass transit systems—whether publicly or privately operated—while continuing to provide safe mobility for essential workers and access to essential services. Read it here.
Does reopening K-12 schools contribute to COVID-19 spread in communities?
Schools are congregate settings from which there is extensive exchange with surrounding communities—that is, students and staff return home every day and interact with people in their household and in the larger community. Reports of superspreader events, outbreaks in other congregate settings—including workplaces and camps attended by both children and adults—and community outbreaks linked with congregate settings have raised concerns about whether the reopening of schools will contribute to the spread of COVID-19 within communities. However, public health officials and pediatric health expert societies concur that in-person education and related programs are fundamental to healthy child development and thus that schools should be open for in-person education as soon and for as long as it is safe. Many schools have reopened fully or are using hybrid remote/in-person learning models. Unfortunately, in the months since many schools have physically reopened, there has been a surge in COVID-19 cases across the United States. In addition, there are now reports of large outbreaks and many smaller outbreaks in schools and some teachers have died from COVID-19. Amid ongoing debate on the risks associated with in-person learning, it is useful to review available evidence on whether COVID-19 in schools contributes to community spread of COVID-19.
A note on institutions of higher education
Like child care and K-12 institutions, institutions of higher education have grappled with decisions about whether and how to reopen. Many have reopened for the 2020-21 academic year. There have been thousands of COVID-19 cases at institutions of higher education. As is the case for K-12 schools, there are questions about the role universities may play in the overall trajectory of the pandemic, and analyses of relevant data have revealed a complex picture. However, there are significant differences between K-12 and higher education environments, including the age of the students, where students live and socialize, student interaction with each other and with the surrounding community, and the fact that many students travel to attend university from outside the surrounding community. In addition, many universities have implemented COVID-19 screening and quarantine policies that differ from what is available or enforced elsewhere. Thus, the data presented here focuses on child care and K-12 educational institutions.
What the data show
One report on factors that have influenced decisions to reopen schools compared school status and COVID-19 infection rates in 191 countries from February through September. By March 31, 96% of countries had partially or fully closed their school systems. Six months later, 64% of countries had at least partially reopened schools. Many countries reported increased COVID-19 incidence compared to when their schools had initially closed. However, data revealed no consistent pattern in the relationship between schools closing or reopening and the rise or fall of COVID-19 infection rates. This suggests that reopening schools may not significantly alter the course of the pandemic. However, there are potential limitations to this analysis. First, the approach to reopening schools has been highly variable between countries, in terms of whether reopening has been partial or full and what infection prevention measures have been in place in schools. Second, other non-pharmaceutical interventions besides the status of schools were not taken into account. Third, the quality and availability of COVID-19 data is not consistent between countries. Fourth, national-level data can disguise patterns at the sub-national level.
The figure below depicts four general relationships—with a country representing each—between school status and COVID-19 incidence within countries.
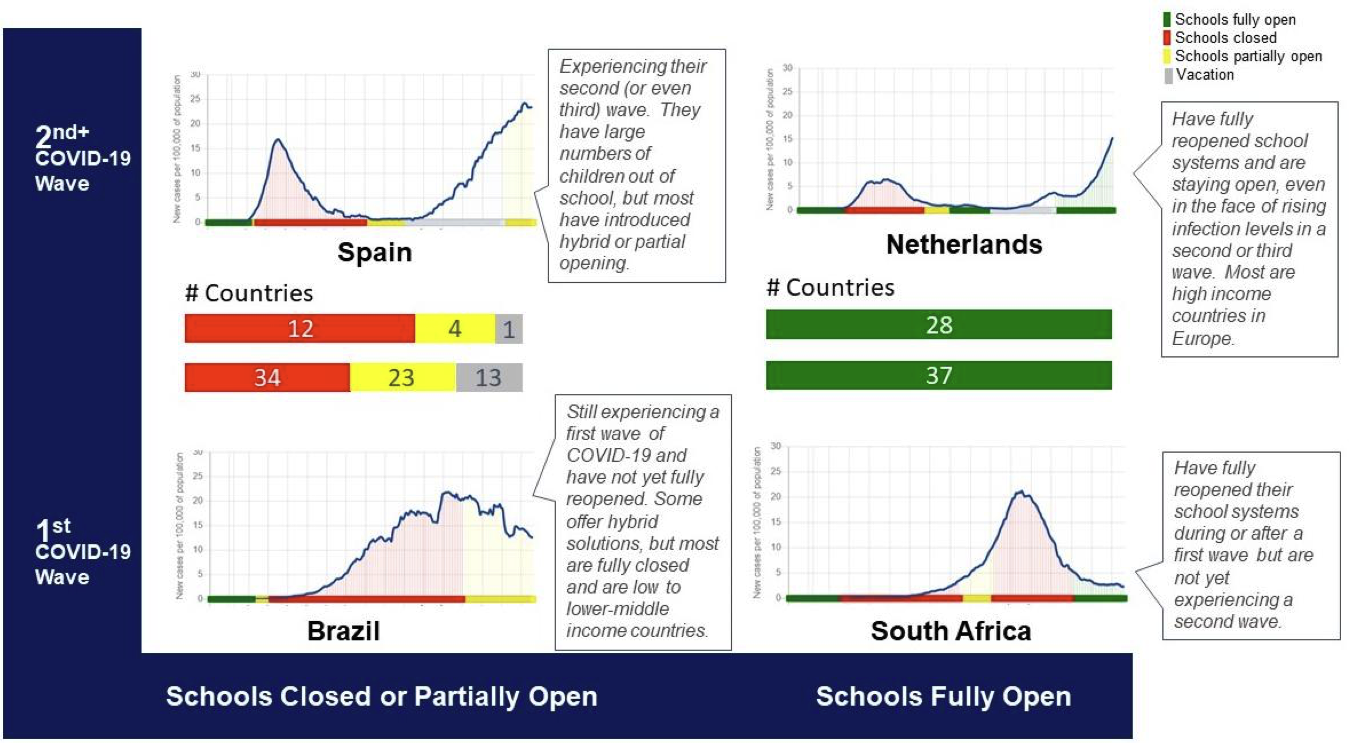
Insights for Education, “COVID-19 and Schools: What We Can Learn from Six Months of Closures and Reopening.”
A modeling study on the effects of non-pharmaceutical interventions on COVID-19 transmission rates using data from 131 countries (reviewed below) showed that closing schools was associated with reductions in transmission rates and that reopening schools was associated with increases in transmission rates. In contrast with the aforementioned report, this analysis took non-pharmaceutical interventions other than the status of schools into account. However, many of the same limitations of the previously mentioned analysis apply, including that data were considered at the national level, that COVID-19 data quality is not consistent across countries, and that neither school-based infection prevention measures nor the degree to which schools were reopened for which age groups were considered. For both analyses, no matter the results, causal relationships cannot be inferred.
Other studies using more granular data—including on local transmission patterns and the results of contact tracing—can inform on the impacts of schools reopening. A rapid review of data on the role of schools in COVID-19 transmission suggested that community transmission is an important driver of the risk of school transmission; that when school-associated outbreaks do occur they typically include fewer than five cases; and that despite high rates of implementation of infection control measures, there is variation in required measures (with rates of mask use lower than rates of other behaviors). A primary conclusion of this review was that the risk of transmission from children within school settings is low. An illustrative study is one from Australia, where 15 adults and 12 children in schools were diagnosed with COVID-19 between January and April. More than 1,000 close contacts were monitored; secondary transmission was documented in four settings and secondary attack rates were higher when the index case was an adult. Although studies have described transmission from children (including a recent household transmission study reviewed in detail below), as we reported in June, children may be less likely to transmit infection than adults. Since June, more evidence has emerged. Systematic reviews have concluded that children are rarely the index cases within households, that children are not the drivers of COVID-19 transmission within schools, and that adults are more likely to infect children than the reverse, though the quality of evidence is generally low.
Another conclusion of this rapid review was that the implementation of infection control measures appears to be important to limiting COVID-19 spread. Two studies from the U.S. illustrate this. In Rhode Island in June and July, COVID-19 cases occurred in 29 (4.4%) of 666 reopened child care programs. Secondary transmission was documented in four programs, one of which had failed to adhere to Department of Health guidelines. At four overnight camps in Maine attended by 642 children and 380 staff, infection prevention and control measures were put in place. Three asymptomatic people tested positive for SARS-CoV-2, the virus that causes COVID-19, after arrival. They were isolated and their cohorts were quarantined. No secondary transmission was identified.
Similar findings emerge in a summary of epidemiologic data from Europe, which suggests that school reopenings have not had a major impact on cases among children or adults within school settings. As of August, a small proportion (<5%) of COVID-19 cases in Europe had occurred among children under 18 years of age. Contact tracing studies have shown that schools were not the source of infection for the majority of children who became ill while attending school. For example, in one study from Germany, among 557 children diagnosed with COVID-19, only 3% of cases were linked to schools (compared with 42% linked to households). Outbreaks in schools have been relatively rare; of 15 European countries that responded to a survey on outbreaks in school settings, nine reported no outbreaks in schools. Of the six countries that reported school-associated outbreaks, only one country reported a cluster of over 10 cases; in the remaining five countries, outbreaks were small and associated with few secondary cases. A non-peer-reviewed analysis of national surveillance data from the United Kingdom showed that there were 67 cases linked to school settings and 30 outbreaks in schools during the month of June (when the number of children attending any educational setting increased from 475,000 to 1,646,000). The number of outbreaks was strongly associated with regional COVID-19 incidence. The probable index case was a staff member in 22 of those 30 outbreaks, and staff were recognized secondary cases more often than students.
The coincidence of Israel’s second pandemic wave with its reopening of schools has received significant attention. Within weeks of Israeli schools reopening in May, outbreaks swept through many schools, resulting in hundreds of cases and thousands of people put under home quarantine. An outbreak of COVID-19 at one high school increased the regional prevalence rate among those aged 10-19 years from 20% to 41%. Of note, neither physical distancing nor mask-wearing was enforced at this school. The relationship between transmission within schools and in the community is difficult to disentangle. It is likely that community transmission contributed to school outbreaks; it is possible that reopening schools exacerbated the situation in the community. An analysis of COVID-19 trends among school-age children in the U.S. showed that among school-aged children, the incidence of COVID-19 increased from March to July and test positivity rates increased from late May to July, plateaued in August and then increased again in September. During March through May, widespread shelter-in-place orders were in effect; in June and July, many communities relaxed mitigation measures; and in August and September, some schools re-opened for in-person learning. Sources of transmission were not analyzed and these data do not show whether transmission within schools contributed to increased incidence. However, the fact that many schools were closed during the months when incidence increased among school-age children serves as a reminder that transmission among school-aged children does not necessarily occur within schools. In fact, the incidence of COVID-19 among adolescents in the U.S. was approximately double the incidence in younger children—potentially due, in part, to the social behavior of adolescents. Many cases among high school students have been linked to parties and other social events. Among school-age children of all ages, a number of cases have been linked to extra-curricular activities such as overnight camps. As mentioned previously, households are also important risk environments, as illustrated by contact tracing studies showing that school-attending children with COVID-19 are more likely to have acquired the infection within their households than at their schools.
What are the risks to teachers and staff within school settings?
We previously reported that children with COVID-19 are less likely to develop severe illness than adults and much less likely to die. However, the comparatively low risk of severe COVID-19 among children must be contextualized within the risk posed to school staff. One analysis that utilized National Health Interview Survey data found that the majority of school staff have risk factors for severe COVID-19 as defined by CDC. Another analysis showed that 42% of school employees are at increased risk of severe COVID-19 and that among school employees, a higher percentage of non-teaching staff (58%) are at increased risk. Both studies showed that millions of Americans with risk factors for severe COVID-19 share a household with a school-age child or school employee.
There are scarce data on the risk of adult infection associated with school settings compared to other settings. In Sweden, the risk of COVID-19 among teachers was not higher than the risk among other adults. A study of 57,000 child care providers across the U.S. conducted in May and June found that continuing to provide child care was not associated with an increased risk of COVID-19. Rather, risk of COVID-19 was associated with the level of transmission in the community and with being a racial or ethnic minority. Over 90% of child care providers reported frequent hand-washing and disinfection of surfaces. Other infection control measures, including temperature checks, physical distancing, and cohorting of children, were frequently implemented as well. The age of the children cared for and the small cohort size (on average, seven children in center-based care and five children in home-based care) may make these findings less generalizable to other educational settings.
How might these data affect decision-making?
School administrators, public health officials and community members across the U.S. have worked hard to implement plans to reopen schools safely. In addition, communities have considered the circumstances that may prompt suspension of in-person learning. Non-prescriptive federal guidance on gauging risk to inform school-related decision-making suggests using indicators of community disease transmission, and different state dashboards include different county-specific data that officials may use to inform decisions about in-person education and risk mitigation. For example, Minnesota presents 14-day case rates, West Virginia presents case rate and test positivity data, and Arizona presents case rate, test positivity and COVID-like illness data. At least one jurisdiction, Franklin County in Ohio, presents absence rates as an indicator that may be used to inform decisions. Increased community transmission in parts of the U.S. has contributed to school closures in a number of locations. Of note, this contrasts with the approach in France, Germany and the United Kingdom, where schools have remained open despite the implementation of lockdowns as COVID-19 cases surge.
Although existing data may not provide definitive answers about how local jurisdictions should handle reopening schools, data can inform and reinforce certain aspects of ongoing work to balance the risks and benefits of in-person education. First, data increasingly suggest that school reopenings are unlikely to contribute significantly to community transmission when rates of community transmission are low and schools have infection prevention measures in place. However, there appears to be a correlation between increased community transmission and the incidence of cases and outbreaks in schools. Thus, monitoring community transmission via the use of several robust indicators—as a number of jurisdictions are doing—is critical to keeping schools and communities safer. Second, although transmission in environments outside schools may be more common, infections acquired within schools can be transmitted to contacts outside school. Thus, tracking and reporting cases associated with schools is critical. Third, schools have not contributed to community outbreaks in the ways that other congregate settings have. Although this may be due in part to reduced transmission from children than from adults, the importance of implementing and enforcing infection prevention and control measures cannot be overstated. Lastly, although the risk of infection among adults exposed to school environments may not be higher than the risk of infection among other adults, adults appear to be at higher risk of infection than children and, if infected, adults are at higher risk of severe disease. Thus, transmission prevention strategies within schools should focus on reducing transmission to and from school staff.
What are monoclonal antibodies and antibody cocktail treatments for COVID-19?
By giving patients recently diagnosed with COVID-19 a dose of targeted antibodies, the goal is the antibodies will inactivate or neutralize SARS-COV-2, the virus that causes COVID-19, and block it from further entering cells, thereby treating the infection. This pathway is similar to how convalescent plasma works in various infections (read more about convalescent plasma in a previous Weekly Science Review here). One major difference is that the antibodies selected to be manufactured in these targeted therapies are thought to have the highest likelihood to be effective and are selected specifically based on results from laboratory research and testing. Another major difference is that the antibodies are produced in a lab, rather than processed from blood donation from recovered patients. Polyclonal antibody treatments, or “cocktails,” contain more than one specialized antibody and may have the added benefit of being able to resist mutations in the virus that could happen over time, which could render one antibody ineffective.
There are two antibody therapies currently under study specifically for fighting COVID-19 infection. These are REGN-COV2—a “cocktail” of two different monoclonal antibodies manufactured by Regeneron—and LY-Cov555, a single monoclonal antibody manufactured by Eli Lilly. In early studies, both had shown that they were able to significantly reduce the amount of virus circulating in infected people (viral load), but to what degree this correlates with clinical improvement is still not clear. On Oct. 26, 2020, Eli Lilly announced that it was stopping its trials of the medication in hospitalized patients as it had failed to show any benefit in these more seriously ill patients. Eli Lilly studies in patients with milder disease are ongoing. Both Eli Lilly and Regeneron have already submitted requests for emergency use authorization (EUA) of their drugs to treat COVID-19.
Other antibody therapies such as tocilizumab—already approved for other disease processes—have also been a part of large trials to identify any potential benefit. This particular monoclonal antibody is designed to target molecules that cause severe inflammation, a process that is thought to significantly contribute to more severe COVID-19 disease. Although some observational studies had shown a potential benefit and reduced mortality in severely ill COVID-19 patients, results from a randomized controlled trial recently published in the New England Journal of Medicine failed to show that tocilizumab prevented moderately ill COVID-19 patients from needing a ventilator or dying.
Antibody treatments are some of the most expensive therapies available, sometimes costing tens of thousands of dollars for a single course of therapy in one patient. Regeneron’s efforts are funded in part by Operation Warp Speed, meaning that under EUA (emergency use authorization) of its antibody cocktail for COVID-19, the treatment will be provided to patients in the U.S. at no cost by the federal government.
Weekly Research Highlights
Note: U.S. CDC also publishes a COVID-19 Science Update
(Lancet Infectious Disease, Oct. 22)
- Researchers used data on country level estimates of R, a measure of the level of disease transmission, from the EpiForecasts project (London School of Hygiene & Tropical Medicine) and analyzed this in a model which also included data on country-specific policies on NPIs from a different database. They looked for any association between changes in R and when NPIs were introduced and lifted.
- R decreased between 3% and 24% 28 days after the introduction of various NPIs as compared to the day prior to introduction. This reduction was statistically significant only for bans on public events with non-statistically significant trends toward reduction in other NPIs. It took a median of eight days to see 60% of the maximum reduction in R. R increased 11% to 25% 28 days after lifting NPIs compared to the day prior to NPI relaxation. This increase was statistically significant only for school reopening with non-significant trends toward increases in R in other NPIs. It took a median of 17 days to see 60% of the maximum increase in R once NPIs were lifted.
- Attention to the time it takes to observe the effect of tightening and relaxing NPIs is necessary to use these mitigating measures most effectively. Policymakers should keep this type of data in mind when planning to maximize safety as they titrate NPIs based on COVID-19 transmission and risk. The immediacy of the effect of varying NPIs differs. In tightening, some NPIs need to be adjusted with early warning signals of increase in disease transmission to allow for the lag it takes for them to have an effect. Similarly, transmission dynamics should be monitored closely for weeks after relaxation of NPIs as the effect of relaxation may take longer to be evident.
Optimal COVID-19 Quarantine and Testing Strategies
(MedRxIv preprint, Oct. 28)
- A mathematical model was used to assess the probability of post-quarantine transmission of COVID-19 using varying quarantine durations and three testing strategies (entry only, exit only, entry and exit) and then compared to the effectiveness of a 14-day quarantine without testing.
- Testing for SARS-CoV-2 was conducted among oil and gas employees from April 11 to Aug. 26, 2020 yielding a total of 4,040 PCR tests to analyze from various times during quarantine. Overall, the duration of quarantine could be substantially reduced when adding a testing strategy to quarantine without increasing the risk of post-quarantine transmission of COVID-19. This was true in both the setting of contact tracing with known COVID-19 exposure, as well as in routine workplace quarantine designed to prevent outbreaks upon entry into or exit from a close contact setting (e.g. offshore rig workers). This data may be extrapolated to other situations where there is “entry” into a higher-risk, close-contact environment from the community such as in a university, with subsequent return to the community. The probability of post-quarantine transmission of COVID-19 was reduced significantly using a quarantine period of nine days with testing on exit from quarantine, and eight days when testing on both entry and exit.
- This data supports the use of testing as an adjunct to quarantine in order to reduce quarantine duration safely while minimizing the risk of an infected asymptomatic individual transmitting disease after quarantine. Authorities will need to make their own decisions about the cost and benefit of testing protocols (e.g. value analysis of eight-day quarantine with two tests versus nine-day quarantine with one test), quarantine periods, and the use of resources such as PCR tests that may be limited in some settings. The goal remains to concurrently maximize safety, minimize risk from COVID-19, and curtail economic disruption.
Repeat COVID-19 Molecular Testing: Correlation of SARS-CoV-2 Culture with Molecular Assays and Cycle Thresholds
(CID, Oct. 27)
- Researchers analyzed retrospective data collected over two months from 2,194 patients and patients under investigation who had received repeat testing for SARS-CoV-2 for a total of 29,686 specimens. For the prolonged positive repeat tests, they then looked for any relationship between cycle threshold values and ability to grow live virus in culture. For the initial negative tests, they used ddPCR to perform confirmatory testing.
- Virus was isolated and recovered in cell culture from tests among four of 29 randomly selected people with prolonged positive repeat testing and was associated with lower Ct counts. This did not correlate with severity of disease. Whole genome sequencing confirmed that the patients were carrying the same virus over time. There was a positive test observed after a negative test in recovering patients in 124 instances. Among a subset of these patients where virus isolation and culture was attempted, none was recovered.
- There were 1,788 patients with repeat negative tests despite symptoms and clinical signs consistent with COVID-19. A subset of these was examined by ddPCR. Eleven of 198 people tested positive by this more refined quantitative analysis, revealing a false negative rate of 5.6%.
- There are limitations to repeat testing for COVID-19, and this strategy should not be used in decision-making about returning to school or work outside of parameters currently recommended by public health authorities. There is a chance that patients with persistent positive test results, especially when they still have symptoms, can continue to shed live virus longer than previously thought. More research is necessary around cycle thresholds and how they relate to contagiousness over time.
Transmission of SARS-COV-2 Infections in Households — Tennessee and Wisconsin, April–September 2020
(MMWR, early release Oct. 30)
- Researchers performed prospective research on household transmission in Nashville, TN and Marshfield, WI from April to September 2020. Once an index case was identified by PCR testing in a household with at least one other person, the other household members were trained and asked to keep symptom diaries, and on how to self-obtain nasal swab or saliva samples for PCR testing daily for 14 days. Samples from the first seven days were tested by PCR.
- A total of 191 household contacts were enrolled for 101 index patients, whose median age was 32 years. Of the contacts, 102 had a positive test for SARS-CoV-2 during the study period, yielding an overall secondary attack rate of 53%. This secondary attack rate was the same for households in which the index patient was over or under 12 years of age. Two thirds of contacts (68 of 102) who became infected developed symptoms within seven days of follow up, although some (27 of 102) were not symptomatic at the time of their first positive test.
- It is possible that the index patient was infected concurrently as another household member who may have been a true asymptomatic index patient. Some infections could have resulted from community exposure outside the household.
SARS-CoV-2 Exposure and Infection Among Health Care Personnel — Minnesota, March 6–July 11, 2020
(MMWR, Oct. 30)
- Since March 6, 2020, the Minnesota Department of Health has been requiring mandatory reporting of health care exposures to SARS-CoV-2, and enrolling health care personnel in monitoring programs. In this study, researchers analyzed data from this reporting and monitoring to shed light on exposures and infections among the state’s health care personnel.
- A total of 21,406 exposures were reported, 5,374 of which were designated to be higher risk requiring quarantine and monitoring. Two thirds of these exposures involved patient care, with the remainder occurring from a coworker, a household member or socially in the community. Within the 14 days following higher-risk exposure, 373 (7%) of health care personnel received a positive SARS-CoV-2 result. Those who had an exposure outside of the patient-care setting had a higher test positivity rate (13%). Those working in group homes had the highest test positivity rate (16%). These workers, along with those working in other congregate settings and long-term care facilities, reported for work more often than those in acute care settings after a higher-risk exposure.
- Some exposures may have been misclassified or not reported properly, and some people who qualified for quarantine and monitoring could not be reached for follow up. The high proportion of exposures outside of patient care settings emphasizes the need for this critical sector of the workforce to practice the 3 W’s at all times. Greater attention is needed to protect residents and staff in long-term care and congregate settings given the higher rates of infection, lower rates of personal protective equipment use, and higher proportion of personnel reporting to duty after an exposure.
Mapping Physical Access to Health Care for Older Adults in Sub-Saharan Africa and Implications for the COVID-19 Response: a Cross-Sectional Analysis
(Lancet, Oct. 1)
- In order to estimate travel time to the nearest hospital or health care facility for adults aged 60 years or older in sub-Saharan Africa at a resolution of 1 km x 1 km, authors assembled a database of health care facilities and their locations by combining data from the OpenStreetMap project (24,571 health care facilities, of which 13,392 were tagged as hospitals) and a geocoded inventory of health care facilities published by the Kenya Medical Research Institute (KEMRI) – Wellcome Trust Research Programme (92,245 health care facilities of which 4,720 were classified as hospitals). Authors estimated the distribution and density of people over 60 years of age using WorldPop data; they estimated travel times using maps of road networks and barriers to travel in combination with estimated speeds on different road types.
- The number of hospitals ranged from 0.07 per 100,000 in Burkina Faso to 11.008 per 100,000 in the Central African Republic. The number of primary health care facilities ranged from 0.03 per 100,000 in Eritrea to 28.053 per 100,000 in Gabon.
- Across sub-Saharan Africa, the proportion of adults aged 60 years and older with an estimated travel time of over six hours to the nearest hospital was 10%, ranging from 0% in Burundi and The Gambia to 41% in Sudan. The proportion of adults aged 60 years or older with a travel time of over two hours to the nearest health care facility was 16%, ranging from 0.4% in Burundi to 59% in Sudan. The median travel time to the nearest hospital for the fifth of adults aged 60 years or older with the longest travel times was about six hours, ranging from 41 minutes in Burundi to about 28 hours in Gabon. In the figure below, the first column of maps shows the population density of adults aged 60 years and older, the second shows the estimated travel time among these adults to the nearest health care facility, and the third column shows travel times for those living in populated areas (defined as areas with at least one adult aged 60 years and older per square kilometer).
Mapping Physical Access to Health Care for Older Adults in Sub-Saharan Africa and Implications for the COVID-19 Response: a Cross-Sectional Analysis
Limitations include the reliance on crowd-sourced data to tag health care facilities, which may explain the high estimate for the number of facilities per population in the Central African Republic. Data on health care facility functionality or readiness to care for COVID-19 patients were not available. This analysis did not consider variations in population vulnerability to COVID-19. The relative importance of other potential barriers to health care access were not considered.
Suggested citation: Cash-Goldwasser S, Kardooni S, Cobb L, Bochner A, Bradford E and Shahpar C. In-Depth COVID-19 Science Review October 10 – 30, 2020. Resolve to Save Lives. 2020 November 5. Available from https://preventepidemics.org/covid19/science/review/








