Data insight:
Using data to assess adherence with public health and social measures can better inform decision-making
Public health and social measures (PHSMs, also known as non-pharmaceutical interventions or NPIs) are an essential component of a COVID-19 response strategy. These measures but should be implemented with care as they can be socially and economically disruptive. WHO has appropriately categorized these measures into personal protective measures such as wearing a mask; environmental measures such as increased cleaning and disinfection of spaces; physical (or social) distancing measures such as shelter-in-place orders; and travel-related measures. Once implemented, it is important to understand adherence with these measures to best understand whether they might be effective. The ongoing COVID-19 pandemic has highlighted existing and new information sources to enable monitoring of adherence. The table below is a non-exhaustive list of some of the data sources that could be used to assess adherence for each measure.
| Category | Measure | Data sources | Notes |
|---|---|---|---|
| Personal protective
| Hand hygiene | Unobtrusive direct observation | Used in healthcare settings |
| Automated/electronic systems for hand hygiene monitoring | Can allow for continuous monitoring and reduce the Hawthorne effect | ||
| Consumption of hand hygiene products (e.g. paper towels, soap, water, hand sanitizer) | Can be applied at population level | ||
| Respiratory etiquette | Unobtrusive direct observation | Harder to measure as events less predictable | |
| Face masks | Unobtrusive direct observation | Can assess in different settings (e.g. community, planes) and visually estimate proper wear | |
| Consumption of face masks | Caveat that consumption does not equal active and proper wear | ||
| Environmental | Surface and object cleaning | Visual assessments of cleanliness | Easy to implement but subjective |
| Direct practice observation | Covert monitoring, more for research | ||
| Evaluating surfaces (includes swab cultures, agar slide cultures, Fluorescent gel/markers, ATP system) | These vary by ease of use, ability to detect pathogen and direct link to cleaning. Mostly for healthcare setting | ||
| Consumption of cleaning supplies and equipment | Indirect general measure | ||
| Physical distancing | Isolation of sick and quarantine of exposed | Health department monitoring | Of isolation and quarantine, will not identify all cases or contacts |
| Use of support provided (e.g. food, shelter) | Can compare to expected use | ||
| Ongoing transmission from identified cases and contacts | Could indicate non-adherence | ||
| School and workplace measures and closure | Violations of order citations | If enforcement stringent | |
| Consumption data (declines in power, water) | Dependent on access to data | ||
| Increases in videoconferencing applications (e.g. Zoom, Teams, school attendance) | Dependent on access to data | ||
| Stay-at-home orders and closure of non-essential services | Mobility data Metrics based on mobility data from private companies (incomplete listing) | Can provide timely and granular information at the local level to assess broad adherence with physical distancing measures. Use mobile devices to identify location, data must be anonymized/aggregate data. Can be compared to epidemiologic information to correlate impact of measures. Not yet clear which measures correlate best with actual adherence to physical distancing recommendations. | |
| Consumption data (e.g. increases in household power, water, trash collection, delivery services) | Dependent on access to data | ||
| Travel-related | Entry and exit screening | Surveys of travelers | Can indicate whether travelers know they were screened |
| Number of travelers screened (vs. expected), those positively identified using screening criteria, referrals, confirmed cases. | Can be used to determine proportion of expected travelers screened and yield | ||
| Internal travel restrictions | Flow monitoring (directly observed, video, toll collection, security checkpoint) | Can be resource intensive | |
| Population movement data (e.g. mobile phone) | Most granular | ||
| Public transit data including mass transit, buses, trains, flights | Number and occupancy, | ||
| Consumption of gasoline | Overall travel indicator | ||
| Border closure (in addition to above) | Immigration/visa monitoring | Identify formal crossings |
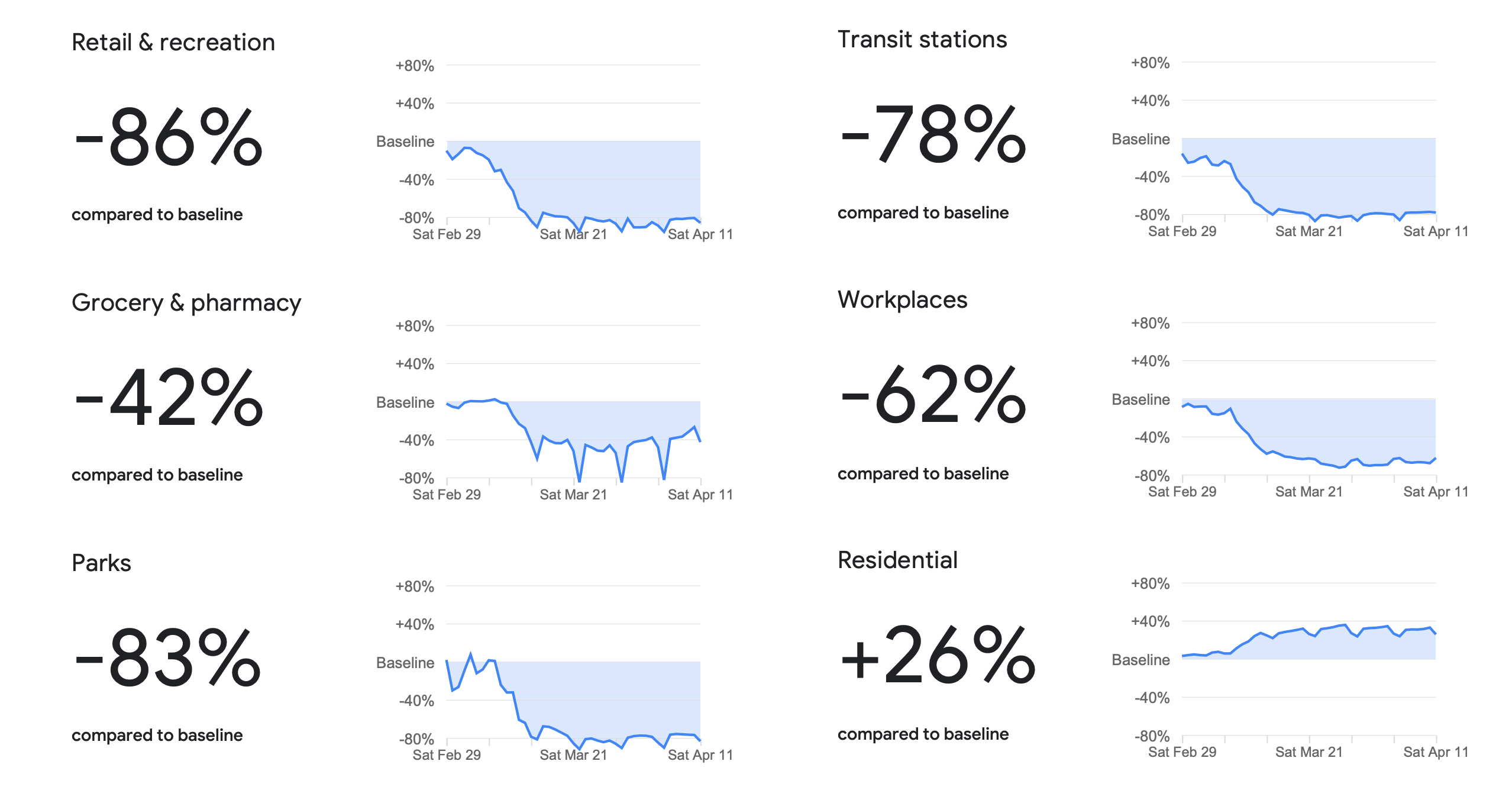
Some of the data sources listed are feasible to examine regularly, others are not. In the context of COVID-19, mobility data in particular has been used to examine the impact of public health and social measures. Many of the companies which have the data, including Facebook, Google and Apple have made it publicly available to researchers during this pandemic. Facebook has developed a Data for Good Mobility Dashboard which provides estimates of relative mobility change and stay-put percentage by US county which can provide important insights. For example, the US state of Georgia has had a 24% reduction in mobility (defined as frequency of travel outside of home area from late February to the current period). However, county-level metrics reveal significant variation in relative mobility reductions. This information can help to better understand which counties have adhered to state- and county-level physical distancing measures, and what additional interventions might be needed.

Source: Facebook COVID-19 Mobility Data Network
Other mobility information available includes specific behaviors such as going to the grocery store or parks, often at a subnational level One can easily see if there are differences in mobility over time as measures are introduced, and across locations as measures are implemented differently.
For example Italy implemented a national stay at home order on March 10, 2020. This included instructions to stay home, limit social contact as much as possible and eliminate non-essential travel. Looking at the Google Mobility data for Italy, one can see dramatic and sustained changes in mobility around this time period.
Source: Google COVID-19 Community Mobility Reports
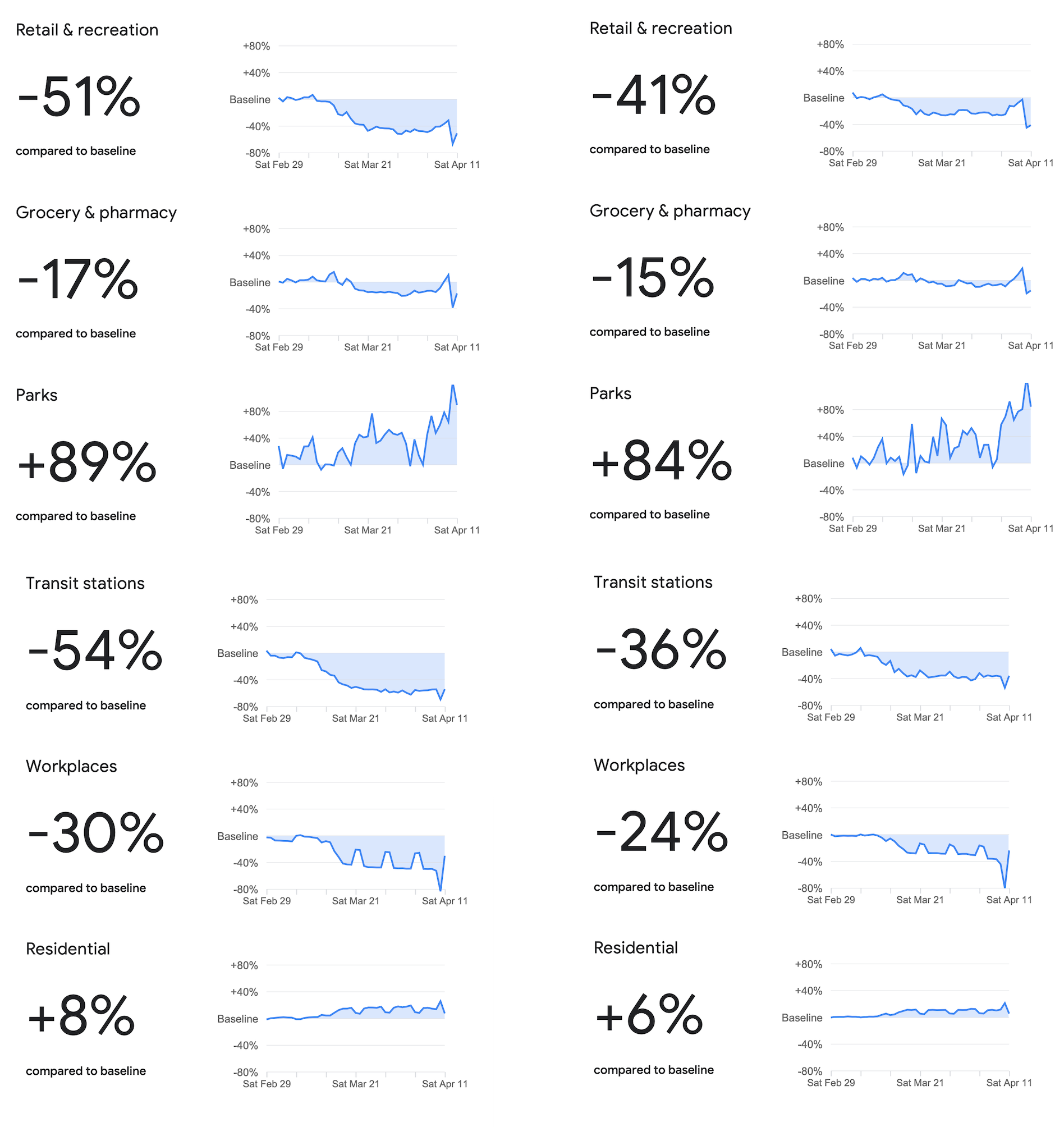
This data can also be used to compare locations. Two European countries which have had different approaches to implementation of public health and social measures are Finland and Sweden. Finland implemented coronavirus restriction in March, including restricting gatherings, closing schools and urging people to stay home. Sweden has less stringent measures in place and has allowed restaurants and business to stay open and has no restrictions on people leaving their home. Comparing the same mobility data shows that despite the highly publicized differences in approaches, there is evidence that changes in behaviors in both countries follow similar trends, but the magnitude of the changes is slightly higher in Finland across all location categories.
Scientists are increasingly using mobility information along with information on disease transmission to assess the impact of public health and social measures. One article in our summary below (Timing of Community Mitigation and Changes in Reported COVID-19 and Community Mobility ― Four U.S. Metropolitan Areas, February 26–April 1, 2020 in MMWR) examined community mobility data as a proxy for physical distancing, and related it to disease transmission information. Last week, we highlighted how the Institute for Disease Modeling in Washington State, USA used estimates of the effective reproduction number to assess whether physical distancing measures and subsequent changes in population mobility are related to reductions in COVID-19 transmission. In that article the authors leverage data from Facebook’s Disease Prevention Maps.
Overall, this type of data can help rapidly identify adherence with physical distancing measures and better inform decision-makers as they consider COVID-19 public health interventions. It should be considered with important information on potentially negative consequences of PHSMs to fully understand their impact. It can also be helpful to contextualize PHSM implementation, as the appropriate set of interventions will vary from one location to another. It is also important to note that population-level trend data can complement but not replace information on essential disease control activities such as rapid isolation of cases and quarantine of contacts.
In depth
SARS-CoV-1 (SARS) vaccines and implications for SARS-CoV-2 (COVID-19) vaccine development
Alleviation of the health and economic burdens of the COVID-19 pandemic may depend on the development of effective vaccines. There are numerous vaccine candidates, many of which have been developed from existing vaccine platforms to target SARS-CoV-2 (the virus that causes COVID-19). There are calls for expedited clinical trial designs that could decrease the time to vaccine rollout. However, some urge caution with respect to timelines as well as optimism, citing concerns that new coronavirus vaccines may cause more severe disease.
The phenomenon of worsened disease severity after immunologic priming has plagued some vaccine development efforts. For example, a vaccine against respiratory syncytial virus (RSV) was associated with enhanced RSV disease and increased hospitalizations, and one particular vaccine to prevent dengue fever was associated with increased risk of death from primary dengue virus infection. Such events can lead not only directly to human suffering but also to a damaging erosion of public trust in vaccines. Immunologic mechanisms have been proposed to explain how some vaccines could worsen disease. In antibody-dependent enhancement (ADE), a vaccine might elicit antibodies that enhance, rather than inhibit, viral entry into host cells. Although ADE has been linked to poor outcomes in cats after vaccination against feline coronavirus (FCoV), there are vast pathogenic differences between FCoV in cats and SARS-CoV-2 in humans. Another mechanism, involving memory cells that mediate inflammatory responses which damage host cells, is potentially more relevant to SARS-CoV-2, based on experience with vaccines against SARS-CoV-1 (the virus that causes Severe Acute Respiratory Syndrome, or SARS).
There are virologic and immunologic reasons why SARS-CoV-1 vaccines may give us insight into what we might expect from vaccines for SARS-CoV-2. First of all, the genome of SARS-CoV-2 is approximately 80% identical to the genome of SARS-CoV-1. Both viruses initiate infections through viral surface proteins that attach to receptors on the surfaces of host cells; immunologic responses to viruses or vaccines can inhibit this attachment. SARS-CoV-1 and SARS-CoV-2 utilize the same host cell receptor, the ACE2 receptor, which is found in human lungs.
The 2003 SARS epidemic had a pronounced global impact, and the scientific response included a focused effort to develop a vaccine. Before the epidemic was ultimately controlled through non-pharmaceutical interventions, two vaccines were evaluated in phase I clinical trials. Both vaccines were well tolerated and resulted in production of neutralizing antibodies (antibodies which appeared likely to counteract the virus). However, no further human trials of SARS vaccines have been conducted, and data on potential adverse events following viral challenge comes from non-human animal studies. Numerous animal models of SARS vaccination and subsequent SARS-CoV exposure demonstrated immunogenicity as well as decreases in viral replication and host tissue damage, without evidence of harm. In some studies, post-vaccination viral challenge was associated with lung damage in mice and hepatitis in ferrets. A major caveat with any non-human animal model is that it may not accurately predict the human response, but these data cannot be ignored. Efforts to develop SARS-CoV-2 vaccine candidates must balance the urgency of this pandemic scenario against the need for rigorous safety testing and monitoring.
Hydroxychloroquine and potential impacts
Early results from small, pilot studies suggest that hydroxychloroquine may lower risk for developing severe COVID-19 or death. Ongoing larger randomized, placebo-controlled trials will determine definitively if hydroxychloroquine provides clinical benefit and is sufficiently safe. Hydroxychloroquine is not currently approved by any major regulatory authority as a COVID-19 treatment or prophylaxis. Clinicians electing to use hydroxychloroquine, particularly in combination with azithromycin, for seriously ill patients should consider the patient’s risk for fatal arrhythmias and ensure their ability to monitor electrocardiogram QT interval and levels of potassium and magnesium on at least a daily basis, or more frequently, if clinically indicated.
FAQs
Red eyes and COVID-19
Some reports have said that COVID-19 can cause reddening (conjunctivitis) or swelling (chemosis) of the white parts of the eyes. According to a nurse from a long-term care facility in Washington State, USA, residents who were sick with COVID-19 all seemed to have red eyes. Notably, Dr. Li Wenliang, who first raised the alarm about coronavirus in December, was an ophthalmologist who believed he contracted the virus from an asymptomatic glaucoma patient. One NEJM study found that 9 (0.8%) of 1,099 COVID-19 patients had “conjunctival congestion”. The WHO-China Joint Mission on COVID-19 estimated the incidence of conjunctival congestion at 0.8%, based on a study in 55,924 laboratory-confirmed cases. Other studies have tested and found SARS-CoV-2 RNA in ocular secretions (1,2). In these studies examining COVID-19 patients, it is important to note they may have had other reasons to have red or swollen eyes. Overall, the American Academy of Ophthalmology notes that based on existing evidence, conjunctivitis is an uncommon event as it relates to COVID-19. Many people can have pink or red eyes, and in the vast majority of them the cause is not COVID-19. Still, these findings are a reminder that the eyes are a mucous membrane, and everyone should protect them as they would their mouth or nose (including not touching them frequently and wearing protection when caring for those who might have COVID-19).
FAQs
Proximity to runners and cyclers
As the SARS-CoV-2 pandemic continues and gyms remain closed, there are questions about whether social distancing recommendations from the Centers for Disease Control and Prevention adequately address the risks of contracting COVID-19 that may be associated with outdoor exercise. Concerns about the safety of outdoor exercise have been intensified by a recent non-peer reviewed study in which a wind tunnel was used to simulate droplet movement around people walking and running in various configurations. The authors suggested that to avoid contact with exhaled droplets, it may be necessary to stay 16 feet away from walkers, 33 feet away from runners, and 65 feet away from cyclists. It has been demonstrated that droplet trajectory is influenced by a variety of factors, including the way droplets are generated and air currents. Indeed, studies have shown that droplets can travel farther than 6 feet. However many virologic factors reduce the risk of droplet-based transmission, and the 6-foot rule is supported by data on other respiratory pathogens. For instance, there must be live virus in a droplet in order to cause infection, and the dose of virus may be an important factor in transmission risk. Longer durations of exposure are associated with COVID-19 infection, and street-passing encounters are often short. Generally, guidelines strive to minimize risk; complete elimination of SARS-CoV-2 transmission risk would require total isolation. As the pandemic continues, the mental health benefits of outdoor time are recognized, and public health recommendations on physical activity cannot be ignored. Overall, those who engage in outdoor physical exercise and those around them should attempt to adhere, at a minimum, with physical distancing guidelines. If physical distancing during exercise is difficult due to public congestion, face masks may help reduce transmission risk.
Articles
Epidemiology
Temporal dynamics in viral shedding and transmissibility of COVID-19
(Nature Medicine, 15 April 2020)
- By examining serial interval, or the time between a primary and secondary case developing symptoms as part of a transmission chain (median 5.2 days, 95% CI 4.1-6.4 days), and incubation period, or the time a person is infected without displaying symptoms (mean 5.2 days from other studies), the authors infer that infectiousness starts prior to symptom onset for a proportion of patients.
- They use quantitative viral testing to support this inference by demonstrating that among 94 lab confirmed COVID-19 patients with 414 throat swabs collected from symptom onset through 32 days after symptom onset, viral loads were highest shortly after symptom onset and decreased towards the detection limit by 21 days after symptom onset.
- The estimated proportion of presymptomatic transmission from this analysis was 44% (95% CI 25-69%).
- More inclusive criteria for contact tracing are recommended to capture potential exposures occurring 2-3 days prior to symptom onset.
Transmission of COVID-19 to Healthcare Personnel During Exposures to a Hospitalized Patient – Solano County, California, February 2020
(MMWR, 17 April 2020)
- The first documented community transmission of COVID-19 in the US occurred in February, 2020, when a patient without travel history or known contacts tested positive after presenting with symptoms of respiratory illness. Prior to diagnosis, the patient had been in contact with 291 HCPs at 2 hospitals.
- At hospital A, 145 HCP with potential exposure were identified from medical records; 121 were confirmed to have had low (27), medium (80), or high (14) risk exposure to the patient. Of these, 43 developed symptoms of COVID-19 and underwent testing, yielding 3 positive results from HCP with high (2) and medium (1) risk exposure. High risk exposures at Hospital A included nebulizer treatments, non-invasive positive pressure ventilation, and endotracheal intubation.
- At Hospital B, where the patient arrived already on a closed-circuit ventilator and was subsequently tested and diagnosed with COVID-19, 146 HCP had exposures, of whom 8 developed symptoms, were subsequently tested, and none tested positive.
- Given the very low rates of community transmission at this time, it is highly likely that these cases were the first cases of occupational transmission to HCP in the US. Additional HCPs who were asymptomatic and were not tested may have also been affected.
Characteristics of Health Care Personnel with COVID-19 – United States, February 12 – April 9 2020
(MMWR, 17 April 2020)
- HCP are at risk for direct and indirect exposures to patients and infectious materials in the workplace, but also in the community and at home.
- Exact rates of likely workplace-related infections among HCPs are unknown due to differences in capturing and reporting this type of data; only a small fraction of cases reported to CDC have included information about whether or not the patient was a HCP.
- HCPs with COVID-19 are less likely to be hospitalized when compared to other patients (8-10% vs 21-31%), and this may be related to the relatively younger average age of HCPs with the illness (42 yrs) or to more complete testing identifying infection with a wider range of symptom severity.
- The number of deaths among these known HCPs with COVID-19 is low, 27, accounting for 0.3-0.6% of cases based on the various data available. More than one third of the deaths (10, 37%) occurred in HCPs over 65 years old.
- HCP should continue to wear the recommended PPE to minimize the risk of exposure and transmission and should exclude themselves from work when ill.
Universal screening for SARS-CoV-2 in women admitted for delivery
(NEJM, 13 April 2020)
- All women who presented for delivery at two hospitals in northern Manhattan were tested for SARS-CoV-2 on admission and assessed for fever and other symptoms of COVID-19.
- Few women had fever or other COVID-19 symptoms on admission, but all who did tested positive for the SARS-CoV-2 virus.
- Among women who were asymptomatic and tested positive on admission, 3 developed fever during their brief hospital stay. One asymptomatic woman who tested negative on admission also developed symptoms and had a positive test three days after the first one.
- These are some of the first data on SARS-CoV-2 infection prevalence among young women without fever or respiratory symptoms during the period when the epidemic was growing exponentially in New York.
- The high proportion of women who tested positive but were asymptomatic and remained so (for a median stay of two days) is important for planning public health measures following the mitigation phase, although the symptom pattern may be different in pregnant women from other infected individuals.
The impact of COPD and smoking history on the severity of Covid-19: A systemic review and meta-analysis
(J Med Virol, 15 April 2020)
- Based on 10 studies with a total of 1,819 patients, COPD was associated with a four-fold greater incidence of severe disease or death (OR 4.38; 95%CI: 2.34-8.20)
- Based on 7 studies with a total of 1,545 patients, smoking was found to increase the risk of severe disease or death two-fold (OR 1.98; 95%CI: 1.29 – 3.05)
Clinical features
Factors associated with hospitalization and critical illness among 4,103 patients with Covid-19 disease in New York City
(MedRxiv preprint, 8 April 2020)
- Age was by far the most powerful predictor of hospitalization. Adults over 75 were 67 times more likely to be hospitalized (OR 66.79; 95% CI: 44.73 – 102.62) and adults 65 – 75 were 11 times more likely to be hospitalized than adults 19-44 (OR 10.91; 95% CI: 8.35 – 14.34).
- Although hypertension and coronary heart disease were more common in hospitalized patients compared to those who weren’t hospitalized (37% v. 11% and 1.8% v. 10%, respectively), neither were significant in the multivariate analysis, suggesting that associations in previous uncontrolled studies may have been driven by age. Tobacco use was also not associated with increased risk.
- Age, obesity and chronic kidney disease were among the few risk factors that emerged as predictors of both hospitalization and severe disease when hospitalized. People with a BMI>40 were six times as likely to be hospitalized (OR 6.20; 95% CI 4.21 – 9.25) and almost twice as likely to have severe disease when hospitalized (OR 1.7; 95% CI: 1.03 – 2.90 when compared to those with BMI<30. People with BMI 30-40 were over four times as likely to be hospitalized (OR 4.26, 95% CI 3.50-5.20) and almost 40% more likely to have severe disease when hospitalized (OR 1.38, 95% CI 1.03-1.85)
- In contrast to other reports, African Americans were not more likely to be hospitalized in this sample. However, in the multivariate analyses, Asians were more likely to be hospitalized (OR: 1.44; 95% CI: 4.21 – 9.25) and to have critical illness (OR 1.91; 95% CI: 1.09 – 3.37). People categorized as “other/multiracial” were also more likely to be hospitalized (OR = 1.99; 95% CI: 1.62 – 2.45).
Management
Chloroquine diphosphate in two different dosages as adjunctive therapy of hospitalized patients with severe respiratory syndrome in the context of coronavirus (SARS-CoV-2) infection: Preliminary safety results of a randomized, double-blinded, phase IIb clinical trial (CloroCovid-19 Study)
(MedRxiv preprint 11 April 2020)
- 81 patients with ARDS and a clinical presentation consistent with COVID-19 illness were randomized to a low-dose (n=40) or high-dose (n=41) therapy group — except for those over 75 who were all placed in the high-dose group. Results from laboratory testing, unavailable at the time of enrollment, confirmed SARS-CoV-2 infection in 62 patients (76.5%).
- An analysis performed on day 6 of the study showed that 10.7% of the low-dose CQ group and 25% of the high-dose group had prolonged QTc interval. 2 patients in high dose group also had ventricular tachycardia preceding death.
- All patients in the study were receiving other medications also known to increase QTc interval including azithromycin and oseltamivir.
- Chloroquine and hydroxychloroquine may pose significant risk to patients at high doses due to QTc interval prolongation that could result in ventricular tachyarrhythmias and sudden cardiac death. Additionally, one patient in this study developed severe rhabdomyolysis, another known adverse effect of chloroquine treatment.
No evidence of clinical efficacy of hydroxychloroquine in patients hospitalised for COVID-19 infection and requiring oxygen: results of a study using routinely collected data to emulate a target trial.
(MedRxiv, preprint 10 April 2020)
- Compared clinical outcomes of patients who received HCQ 600 mg daily within 48 hours of admission and those who did not. Included confirmed COVID-19 patients between 18-80 years of age who had an oxygen requirement upon admission to one of 4 French hospitals. Excluded those who received treatment with any other experimental drug (including steroids) within 48 hours of admission, and those with ARDS or other end-organ failure at admission.
- Outcomes were analyzed based on a propensity score model with inverse probability of treatment weighting to approximate an unbiased treatment effect of HCQ.
- Among the 181 patients eligible for analysis, 84 received HCQ within 48 hours of admission and 97 did not (although 8 of those did receive HCQ later). Median age 60; 71% men, and initial disease severity was balanced between groups. In the HCQ group, 17 (20%) received concomitant azithromycin and 64 (76%) received concomitant amoxicillin-clavulanate.
- The duration of symptoms prior to admission was 7 days. Most participants had evidence of a significant inflammatory response at admission (CRP > 40 mg/l)
- Comparing outcomes in HCQ versus non-HCQ treatment groups:
- ICU transfer or death within 7 days: 16 (20.5%) versus 21 (22.1%); RR 0.93 (95% CI 0.48–1.81).
- Death: 3 (2.8%) versus 4 (4.6%); RR 0.61 (95% CI 0.13–2.90)
- ARDS: 24 (27.7%) versus 23 (24.1%); RR 1.15 (95% CI 0.66–2.01)
- 8 (9.5%) of patients experienced ECG modifications requiring HCQ discontinuation at a median of 4 days.
Prevention
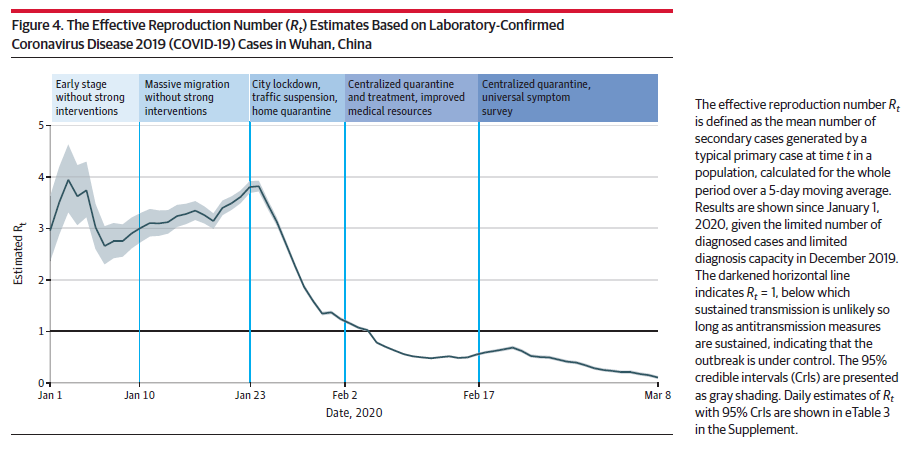
Association of Public Health Interventions with the Epidemiology of the COVID-19 Outbreak in Wuhan, China
(JAMA, 10 April 2020)
- Based on 32,583 laboratory confirmed cases reported in the first three months, daily estimates of confirmed case rates were calculated for 5 intervals, corresponding to distinct stages of the outbreak and response:
- 08 DEC to 09 JAN—No intervention: 2.0 confirmed cases/ million population/ day
- 10 JAN to 22 JAN—massive human movement (New Year holiday) and no intervention: 45.9 confirmed cases/ million population/ day
- 23 JAN to 01 FEB—city lockdown, travel/ traffic restrictions, home quarantine: 162.6 confirmed cases/ million population/ day
- 02 FEB to 16 FEB—centralized quarantine and treatment strategy added: 77.9 confirmed cases/ million population/ day
- 17 FEB to 08 MAR—universal symptom surveys added: 17.2 confirmed cases/ million population/ day
- Estimated Rt fluctuated > 3.0 before 26 JAN; decreased to < 1.0 after 06 FEB; and to < 0.3 after 01 MAR (see figure 4 below).
- Although data are entirely ecological, the fastest decline followed the city lockdown, travel and traffic restrictions, and home quarantining of cases and contacts initiated on 23 January. Stacking multiple NPI components in this way may have been key.
- Overall, the daily case rate was 3 times higher in health workers than in the general population.
- An accompanying editorial suggests that nowcasting parameters such as daily infection rate and Rt from surveillance data could help guide decisions about deploying NPIs elsewhere.
MMWR. Timing of Community Mitigation and Changes in Reported COVID-19 and Community Mobility – Four U.S. Metropolitan Areas, February 24 – April 1, 2020
(MMWR, 17 April 2020)
- In each jurisdiction, the declaration of a state of emergency (state, local, or both) was followed by incremental additions of other community mitigation policies including limits on mass gatherings, school closures, business restrictions, and stay-at-home or shelter-in-place orders. The city of New Orleans added a curfew to its most highly affected areas.
- Following the addition of each mitigation policy over time a smaller percentage of people were observed leaving home (80% on Feb 26 vs 42-61% by Apr 1).
- Though a temporal relationship could be established between implementation of mitigations strategies and decreased community mobility as well as decreased percent change in cumulative case counts, the authors acknowledge that this is an association and cannot prove a causal relationship.
- Heightened community awareness of the pandemic, as well as increased personal mitigation measures such as handwashing, mask wearing, and personal protection, could also contribute to improved control.
Suggested citation: Cash-Goldwasser S, Jones SA, Bochner A, Cobb L and Frieden TR. In-Depth COVID-19 Science Review April 11 – 17, 2020. Resolve to Save Lives. 2020 April 20. Available from https://preventepidemics.org/covid19/science/review/