The latest on COVID-19 treatments: evidence, approvals, guidelines and the implications of new variants.
What the natural history of COVID-19 means for treatment possibilities
When a person has COVID-19, the course of illness includes two main processes. Early in the course of illness, the disease is driven by the replication of the SARS-CoV-2 virus. The immune system begins working to clear the infection and aid recovery, but as time passes, in some people the immune response may become dysregulated, or overactive, causing damage to the lungs and other organs and contributing to the development of severe disease. COVID-19 therapies can generally be divided into two types: those that target the virus (antivirals) and those that target the immune system (immunomodulators). Treatments of both types have been approved and recommended for use in COVID-19 patients. However, a number of viruses, including influenza, can develop mutations that render certain treatments less effective, and antiviral therapies might be less effective against emerging SARS-CoV-2 variants. In contrast, because immunomodulators target the immune system, their effectiveness is less likely to be affected by new viral variants.
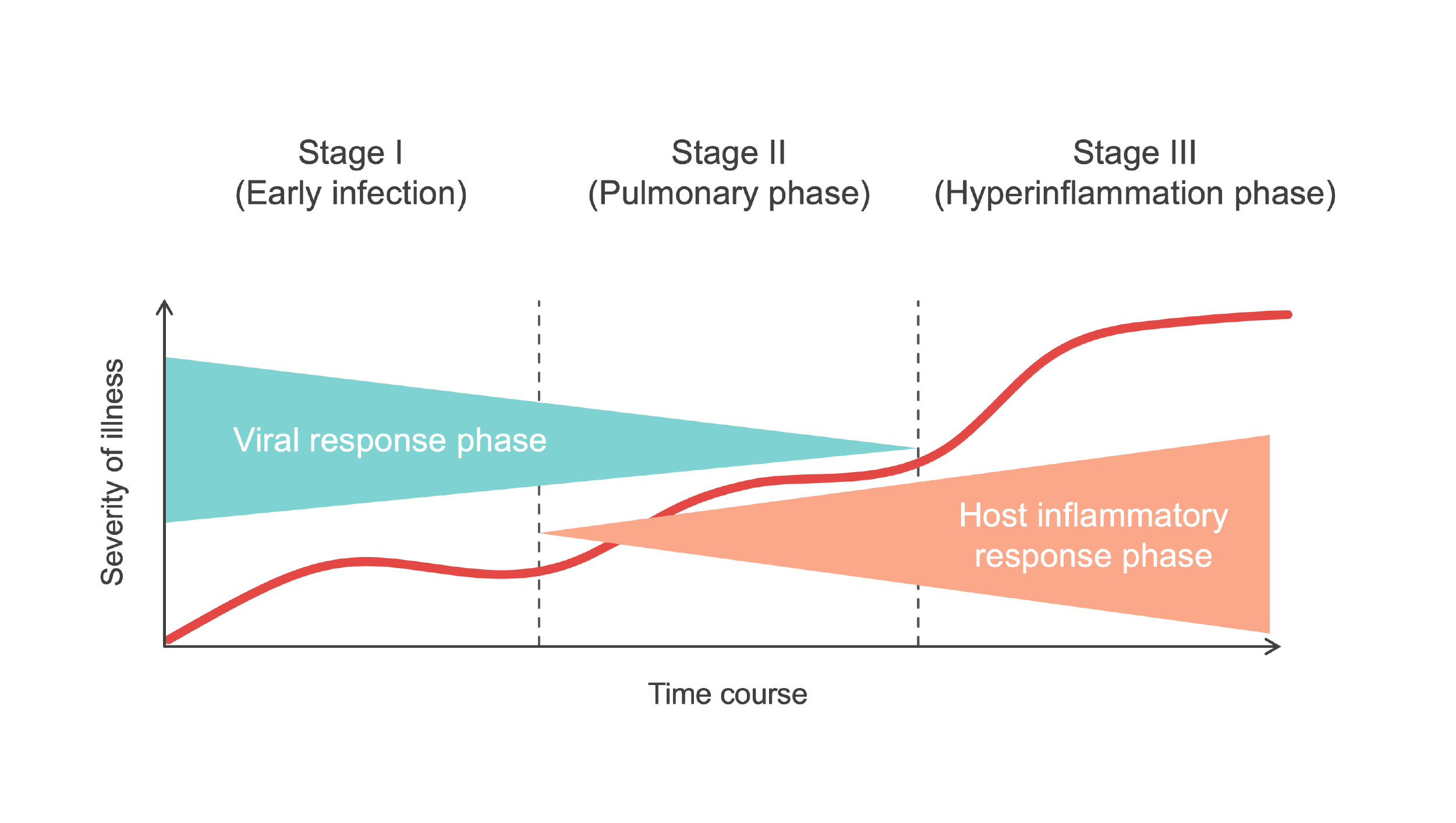
Source: Medmastery
What processes determine whether a drug will be made available to treat COVID-19 patients and whether it will actually be used?
After a potential COVID-19 treatment has been developed and tested, whether or not it will be used is determined by national regulatory processes, the recommendations of expert health organizations and accessibility. In the U.S., the use of pharmaceuticals is regulated by the Food and Drug Administration (FDA); typically, before a new drug can be used, the FDA must issue approval. During a public health state of emergency such as the COVID-19 pandemic, a treatment that has not been approved to treat a specific condition may be authorized as treatment through an Emergency Use Authorization (EUA). An EUA authorizes use to “treat or prevent serious or life-threatening diseases or conditions caused by…threat agents where there are no adequate, approved and available alternatives.” In determining whether to issue an EUA, the FDA evaluates whether the known and potential benefits of the product outweigh the known and potential risks. For drugs that are already on the U.S. market, an EUA may not be needed. A drug called remdesivir is the only treatment that the FDA has approved for COVID-19. Countries and regions outside the U.S. have their own regulatory processes.
Approval or authorization makes drugs available to health care providers to decide whether the treatment is appropriate for a COVID-19 patient. Decisions to use approved or authorized treatments are influenced by health care providers’ interpretations of the evidence and their clinical experience along with best practice guidelines formulated by panels of experts. In the U.S., the National Institutes of Health (NIH) and the Infectious Diseases Society of America have issued COVID-19 treatment guidelines based on efficacy and safety data. NIH guidelines include three types of recommendations: recommendations for treatments, recommendations against treatments and guidance that there is insufficient evidence to recommend for or against a treatment. Currently, the NIH recommends several COVID-19 treatments based on severity of disease; the guidelines are updated as new evidence becomes available. For example, on Feb. 23, 2021, NIH guidelines were expanded to include two new monoclonal antibody treatments for non-hospitalized people with mild or moderate COVID-19 (yet to be reflected in the diagram below, which was updated Feb. 11).
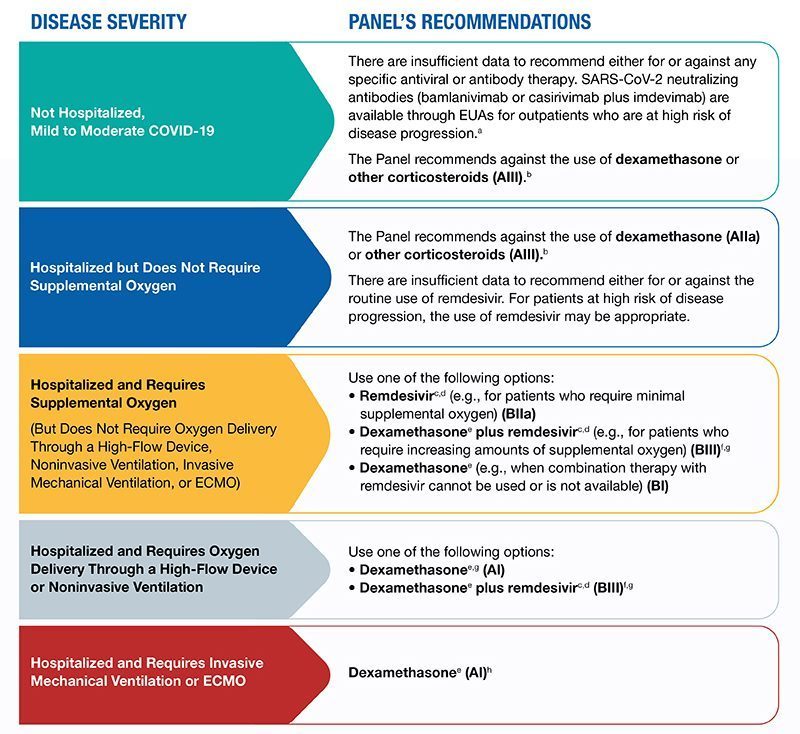
Source: NIH
Outside of the U.S., many countries have their own advisory bodies that issue treatment guidance for a range of diseases including COVID-19. For example, COVID-19 treatment guidance is available from the South African National Institute of Communicable Diseases and the U.K. National Institute for Health and Care Excellence. In addition, the World Health Organization (WHO) issues COVID-19 treatment recommendations that are used around the world. Similar to the NIH, WHO recommends for or against treatments, rating recommendations as “strong” or “conditional” based on the strength and quality of the evidence
Therapies that target the virus (antivirals)
Here we describe three antiviral treatments that are being used to treat COVID-19 patients. COVID-19 antiviral treatments differ from each other in terms of their mechanism of action, strength of evidence to support their use, approval status and recommendations, patterns of use, how accessible they are and what the level of concern is that new SARS-CoV-2 variants may be treatment-resistant. Our three selected treatments differ from each other on several of these parameters. These are far from the only examples of therapies that may provide benefit to COVID-19 patients. There are also a number of drugs with antiviral properties in development, and several therapies developed to treat other diseases have been assessed for use in COVID-19 patients or are currently being tested in clinical trials.
1. Antibody therapies
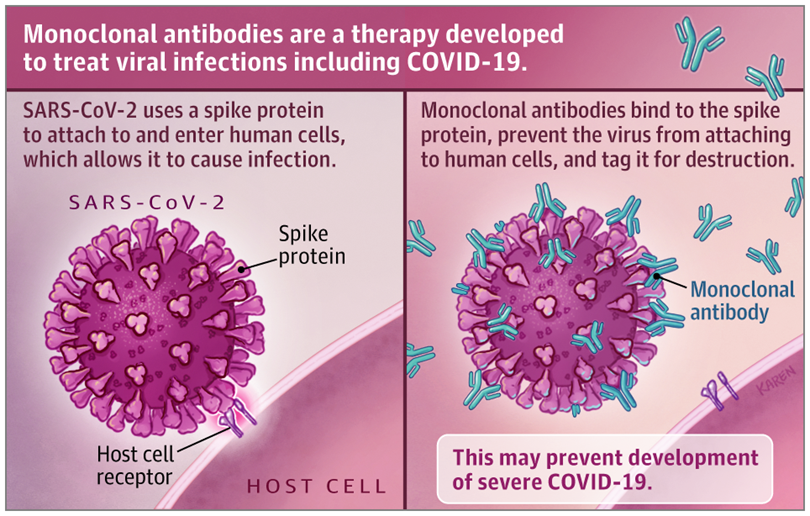
As we previously wrote, antibodies are part of our immune system, produced by immune cells called B cells. Antibodies work against viruses by binding to a protein on the surface of viral particles, which blocks the entry of the virus into human cells. Viruses that cannot enter human cells are unable to replicate. Administering antibody therapy obviates the need to wait for a person to generate antibodies themself, which takes days.
There are several forms of antibody therapy used to treat or prevent infectious diseases, including convalescent plasma and monoclonal antibodies. Plasma is part of the blood that contains antibodies; convalescent plasma is taken from people who have previously been exposed to, or vaccinated against, a disease. The antibodies in convalescent plasma are polyclonal, meaning they have been produced by different lines of B cells and they bind to different targets (e.g., different parts of a virus). Monoclonal antibodies, in contrast, are produced by one line of B cells and have singular, specific affinity for one target. Monoclonal antibodies are produced in a lab by cloning antibodies with specific targets that have been obtained from people, mice or an antibody library. In the case of COVID-19 treatments, the targets are the proteins on the surface of the SARS-CoV-2 virus.
Monoclonal antibody therapies have been used for decades to treat a variety of conditions; approximately 500 monoclonal antibody therapies have been developed around the world and 80 have been approved in the United States. Examples include rituximab (Rituxan), which is used to treat rheumatoid arthritis and other autoimmune conditions as well as certain types of cancer, and adalimumab (Humira) which is used to treat inflammatory bowel disease and other autoimmune conditions. With regard to infectious diseases, monoclonal antibody products can be used to treat Ebola and to prevent respiratory syncytial virus. Because COVID-19 is driven by replication of the virus early in the course of infection, if given early monoclonal antibody treatment may prevent further (severe) disease.
Source: JAMA
New and existing authorizations and guidelines
The monoclonal antibody products to treat COVID-19 that are furthest along in clinical trials were created by the pharmaceutical companies Eli Lilly and Regeneron. Eli Lilly’s monoclonal antibody products, bamlanivimab and etesevimab, and Regeneron’s products, casirivimab and imdevimab (produced in a “cocktail” known as REGN-COV2), all have affinity for the spike protein of SARS-CoV-2. The spike protein is what the SARS-CoV-2 virus uses to attach to the ACE-2 receptor on human cells to gain entry. The antibodies bind to the virus to block viral entry into human cells.
The FDA granted EUAs for bamlanivimab and REGN-CoV2 in November 2020 and granted an EUA for the combination of bamlanivimab and etesevimab in Februrary 2021. These EUAs were based on data from randomized controlled trials conducted in outpatients with mild or moderate COVID-19. In the REGN-CoV-2 trial, 6 of 93 patients (6%) in the placebo group and 6 of 182 patients (3%) had at least one medical visit due to COVID-19. Data on bamlanivimab alone or with etesevimab come from the BLAZE-1 trial. For bamlanivimab alone, 5 of 309 patients (1.6%) in the bamlanivimab group and 9 of 143 patients (6.3%) in the placebo group were hospitalized or visited an emergency room. For bamlanivimab plus etesevimab, there was 70% reduction in COVID-19-related hospitalizations or death from any cause among the 518 participants who received the trial therapy, compared with 517 participants who received placebo; there were no deaths in the treatment arm and 10 deaths (2% of participants died) in the placebo arm. On Feb. 23, NIH guidelines were updated to include a recommendation for the use of bamlanivimab plus etesevimab, in combination, for the treatment of outpatients with mild to moderate COVID-19 who are at high risk of severe disease. NIH guidelines state there are insufficient data to recommend for or against the use of bamlanivimab alone, or for or against the use of the combination of casirivimab plus imdevimab (REGN-COV2) to treat COVID-19. However, the NIH recommends against use of monoclonal antibody treatments in hospitalized patients.The trials in which monoclonal antibody products were given to hospitalized patients have been halted due to lack of efficacy or safety concerns upon analysis of interim results; the FDA has not authorized use of these products in hospitalized patients.
Convalescent plasma is generally expected to work in a similar way to monoclonal antibody therapy: antibodies attach to the virus and block cellular entry. However, the antibodies in convalescent plasma are not targeted to a single viral protein and the dose of antibodies can be difficult to control and study. There is an EUA for convalescent plasma from the FDA, though data to support its use are mixed; studies have found both benefits and no benefits in hospitalized COVID-19 patients and several randomized controlled trials have been unable to fully assess treatment impact due to low enrollment. Analyses of observational data from the large “Expanded Access to Convalescent Plasma for the Treatment of Patients with COVID-19” program suggest that the risk of death may be reduced among non-intubated patients who receive convalescent plasma from people with high levels of antibodies within three days of their COVID-19 diagnosis. A recent meta-analysis of 10 randomized clinical trials comparing COVID-19 patients treated with convalescent plasma to those who received placebo or standard of care found that treatment with convalescent plasma was not associated with a decrease in death or improvements in clinical outcomes such as length of hospitalization and need for ventilation. The NIH states there are insufficient data to recommend for or against the use of convalescent plasma to treat COVID-19, while WHO does not mention convalescent plasma in its treatment guidelines.
New variant concerns
Several circulating SARS-CoV-2 variants of concern have mutations that affect the spike protein that is targeted by leading monoclonal antibody therapies. Researchers have conducted laboratory studies to discern whether monoclonal antibody therapies are less effective in treating cases caused by the B.1.1.7 variant (first recognized in the U.K.) or by the B.1.351 variant (first recognized in South Africa). In one laboratory-based study (not yet peer reviewed), monoclonal antibody regimens in clinical use or under study for clinical use were less effective at binding to the B.1.351 variant. In contrast, the B.1.1.7 variant was not resistant to binding by these monoclonal antibody regimens. Most leading monoclonal antibody therapies target a part of the spike protein called the receptor binding domain. The B.1.351 virus has mutations that affect the shape of the receptor binding domain, which likely explains the reduced binding ability of monoclonal antibodies. One of these mutations in the receptor binding domain, E484K, also occurs in the P.1 variant (first identified in travelers from Brazil), suggesting that some monoclonal antibody therapies also may be less effective against this variant. Therapies that contain more than one type of monoclonal antibody (sometimes called “cocktails”), such as REGN-COV2, may be more effective against variants. For example, the imdevimab component of REGN-COV2 retained its potency against the B.1.351 variant, whereas the activity of casirivimab was reduced. With two complementary antibodies in one treatment, even if one has reduced potency there is less risk of the cocktail losing efficacy because the virus would need to mutate in multiple distinct locations to evade both antibodies.
The efficacy of convalescent plasma may also be reduced if infection is caused by a new SARS-CoV-2 variant. Laboratory evidence suggests that convalescent plasma from people who had COVID-19 before the emergence of new SARS-CoV-2 variants has reduced neutralizing activity against emerging viral variants including B.1.1.7 and B.1.351. For example, a study of 35 convalescent plasma samples collected from 17 individuals found that the E484K mutation in the spike protein receptor binding domain (characteristic of variants B.1.351 and P.1) could reduce viral neutralization by convalescent serum by more than tenfold. Evidence of reduced activity of convalescent plasma against emerging viral variants may have another important implication: that people previously infected with COVID-19 are susceptible to reinfection with a new variant. If the antibodies in convalescent plasma cannot neutralize a SARS-CoV-2 variant, the person from whom the plasma was obtained may not be protected against reinfection with a variant. There have been reports of reinfection with new variants, including a person in Manaus, Brazil, who was reinfected with the P.1 variant just eight days after a positive antibody test for SARS-CoV-2. Data from the placebo arm of the Novavax vaccine trial in South Africa suggest that people with SARS-CoV-2 antibodies from infections that occurred before widespread emergence of the variant B.1.351 may be as likely to be infected with the variant as those with no evidence of prior infection.
Another potential concern about antibody treatments is that they may fuel the emergence of new viral variants. If the SARS-CoV-2 virus is exposed to neutralizing antibodies, it may evolve to evade those antibodies. Indeed, there is evidence that treatment with convalescent plasma in the setting of chronic SARS-CoV-2 infection can contribute to the emergence of variant mutations. To prevent emergence of new strains resistant to treatment, research suggests that it is important that treatment cocktails contain antibodies that target different parts of the virus and to ensure that selected targets are not collectively modified by single mutations.
Current status and future directions for monoclonal antibodies
In the U.S., there are reports that despite increasing availability, use of monoclonal antibodies has been limited. There are several likely explanations for this. Monoclonal antibodies can only be administered intravenously and are recommended for COVID-19 patients who are not hospitalized. Patients are also likely to benefit most if therapy is given early in the course of infection. It may be difficult to identify non-hospitalized COVID-19 patients who are at risk of severe disease and to organize the administration of intravenous therapy—while managing the risk of infection to others—all within a few days of diagnosis. Logistical challenges are augmented by mixed supporting evidence, leading to tempered enthusiasm for these products among physicians. In addition, monoclonal antibody products are very costly (although costs to patients in the U.S. are currently covered under Operation Warp Speed). The high price and logistical challenges limit accessibility, particularly in low-income settings.
There may be a role in the future for monoclonal antibodies to prevent disease among those at high risk of disease or poor outcomes (such as unvaccinated people exposed to SARS-CoV-2 who are at high risk of severe disease). Indeed, for a number of infectious diseases, medications or vaccinations that reduce the risk of infection among those who have been exposed are important to transmission prevention activities. Study results posted by the pharmaceutical companies Eli Lilly and Regeneron have suggested that monoclonal antibodies given to exposed close contacts may reduce transmission of SARS-CoV-2 among nursing home residents and household contacts of COVID-19 cases. However, these trials were small and the data have not been peer-reviewed. In the future, the use of monoclonal antibodies to prevent infection after exposure must be reconciled with current guidance that COVID-19 vaccination should be deferred for 90 days after receiving antibody products.
| Therapeutic agent | General mode of action | FDA authorization | NIH recommendation | WHO recommendation | Route of administration | Cost |
|---|---|---|---|---|---|---|
| Convalescent plasma from COVID-19 survivors | Antiviral | EUA | Cannot recommend for or against | Not mentioned | Intravenous | High |
| Ivermectin | Antiviral | Approved to treat non-COVID-19 conditions. No approval or EUA to treat COVID-19. | Cannot recommend for or against | Not mentioned | Oral | Low |
| Monoclonal antibodies targeting SARS-CoV-2 | Antiviral | EUAs for:
| Bamlanivimab+ etesevimab recommended for outpatients with mild or moderate COVID-19 | Not mentioned in current guidance; draft in progress | Intravenous | High |
| Remdesivir | Antiviral | Approved | Recommended for inpatients with severe COVID-19 | Cannot recommend for or against | Intravenous | High |
| Monoclonal antibodies targeting IL-6 (e.g., tocilizumab) | Immunomodulator | Approved to treat non-COVID-19 conditions. No approval or EUA to treat COVID-19. | Cannot recommend for or against | Not mentioned | Intravenous | High |
| Steroids (e.g., dexamethasone) | Immunomodulator | Approved to treat non-COVID-19 conditions. No approval or EUA to treat COVID-19. | Recommended for inpatients with severe COVID-19 | Recommended for inpatients with severe COVID-19 | Intravenous or oral | Low |
2. Remdesivir
Remdesivir was originally developed as a potential treatment for Ebola during the 2013 epidemic in West Africa, and was found to have in vitro activity against SARS-CoV-2 early in the COVID-19 pandemic. It mimics a molecule that viruses use when manufacturing genetic copies of themselves and thereby interferes with viral replication; when a virus’s gene transcription protein (polymerase) attempts to incorporate remdesivir into a new genetic strand, the strand is terminated. Since remdesivir has specific antiviral activity, it is theoretically possible that new SARS-CoV-2 variants could become resistant; for other viruses mutations in viral polymerase genes confer resistance against some treatments. Although the emergence of SARS-CoV-2 variants with resistance to remdesivir is possible, the notable mutations in newly emerged SARS-CoV-2 variants occur in the genes encoding for their spike proteins and there is currently no reason to suspect resistance to remdesivir.
Randomized trials on the use of remdesivir in patients with COVID-19 have yielded mixed results, which has contributed to lack of consensus from expert groups on whether to recommend the treatment. The largest so far were the Adaptive Covid-19 Treatment Trial (ACTT-1) and Solidarity trials. The ACTT-1 trial randomized 1,062 hospitalized COVID-19 patients to receive either remdesivir (541 people) or placebo (521 people). Results showed a reduction in time to clinical improvement (7 days for remdesivir vs. 9 days for placebo; recovery rate ratio 1.45; 95% CI, 1.18–1.79) and, among patients requiring oxygen but not ventilation, mortality in the remdesivir arm was 4% versus 12.7% in the placebo arm (Hazard Ratio 0.30, 95% CI 0.14-0.64). The Solidarity trial included 11,330 adults, 2,750 of whom were assigned to receive remdesivir. Interim analysis of trial data found no evidence that remdesivir had a beneficial effect on hospitalized patients with COVID-19 in terms of duration of hospital stay, initiation of ventilation or mortality. There are a number of reasons why different clinical trials might produce different results. If studies include patient populations that differ demographically, by duration of illness or by disease severity, different outcomes may be observed. For trials of antiviral therapies, the time between illness onset and administration of therapy may be critical. Depending on the current or local standard of care, administration of other treatments concurrently with the trial drug also may differ between trials and affect results. For example, approximately one-quarter of patients in the ACTT-1 trial received steroids, while nearly half in the Solidarity trial did. Also, results may be affected by study design, including whether the trial is placebo-controlled (ACTT-1) or not (Solidarity). More data on remdesivir from other trials, including the RECOVERY and DISCOVERY trials, are forthcoming.
Remdesivir is currently the only therapy approved by the FDA to treat COVID-19. NIH guidelines recommend the use of remdesivir in hospitalized COVID-19 patients requiring supplemental oxygen. On the other hand, WHO recommends against the use of remdesivir, citing insufficient evidence of improved patient-important outcomes. Like antibody therapies, remdesivir is a costly medication that can only be administered intravenously. In addition, the benefits of treatment are likely greatest when administered earlier in the course of treatment, which may explain the mortality benefit in those who need oxygen but not ventilation.
3. Ivermectin
For decades, ivermectin has been used to treat human diseases caused by parasites including onchocerciasis (also known as river blindness), soil-transmitted helminthiasis (commonly known as intestinal parasites) and scabies. It is also used in veterinary medicine. Studies have shown that ivermectin has activity against several viruses in the lab, including SARS-CoV-2. Although concentrations used in the laboratory studies were much higher than what might be achievable in the human body, the treatment is of global interest because it is cheap and administered orally as a pill. This would fill a critical gap since there are currently no other treatments for outpatients with COVID-19 that are affordable and easy to administer.
Observational and clinical trials have evaluated the use of ivermectin for the prevention and treatment of COVID-19. However, the results of studies have not been consistent and no large randomized clinical trial has delivered clear results that ivermectin improves the outcomes of people with COVID-19 or effectively prevents disease. A systematic review and meta-analysis, including 18 randomized controlled trials with results from 2,282 patients, showed that ivermectin was associated with faster viral clearance and shorter duration of hospitalization compared to control treatments. In six randomized controlled trials on ivermectin to treat moderate or severe COVID-19, there was a 75% reduction in mortality with 14/650 (2.1%) deaths among those on ivermectin; 57/597 (9.5%) deaths in controls. However, studies varied widely in terms of design, patient characteristics, standards of care, concurrent administration of other therapies, and in the dose of ivermectin and treatment duration across trials. It can be difficult to draw conclusions from trials that were not conducted in a standardized way and the authors concluded that larger randomized trials are needed before results are sufficient for review by regulatory authorities.
Despite the lack of conclusive evidence, there has been significant global interest in the use of ivermectin and pressure on regulators to issue authorizations. The FDA, however, has warned against taking ivermectin to treat or prevent COVID-19. Ivermectin is generally safe and well-tolerated when taken at doses recommended in appropriate clinical scenarios, but when used inappropriately, it can cause serious harm. In addition, overuse when there is no benefit could cause harm at the population level by facilitating the emergence of ivermectin resistance in parasites. NIH guidelines state that there are insufficient data to recommend for or against the use of ivermectin for COVID-19. The Africa Centres for Disease Control and Prevention also issued a statement on the lack of proven efficacy or safety data on ivermectin as a treatment for COVID-19. Despite the lack of evidence and these warnings, ivermectin is being prescribed in many parts of the world including in countries in Latin America and Africa. Trials using ivermectin to treat COVID-19 are ongoing and new evidence may soon clarify whether ivermectin can help treat or prevent COVID-19. Although ivermectin has been found to have antiviral properties, the mechanism of action may be the inhibition of human proteins that viruses hijack to enhance infection. Therefore, if proven effective, it is theoretically unlikely that mutations found in SARS-CoV-2 variants would have an effect on the efficacy of treatment.
Therapies that target the immune system (immunomodulators)
Here we review two types of immunomodulator therapy. The first type, steroids, have become the standard of care for patients with severe COVID-19. The second type, monoclonal antibody therapy that interferes with immune system signaling, has recently garnered attention because the results of new trials have provided mixed evidence that this type of treatment can reduce the risk of death among COVID-19 patients. Early success with steroid treatment focused attention on immunomodulators for the treatment of COVID-19; a number of other immunomodulators have been or are being tested in COVID-19 patients. Since immunomodulators do not act directly on the virus but rather on components of the human immune system, it is not anticipated that their efficacy would be reduced when used to treat COVID-19 cases caused by new SARS-CoV-2 variants.
4. Dexamethasone and other steroids
Steroids are hormones produced by the adrenal gland that are critical components of many physiological processes, including the immune response. Synthetic derivatives of steroid hormones, including dexamethasone, prednisone and hydrocortisone, are used for their anti-inflammatory effects to treat many inflammatory, allergic and immunologic diseases. For decades, steroids have been suggested, tested and used to treat a number of infectious diseases including severe pneumonias, as well as Acute Respiratory Distress Syndrome (ARDS), a life-threatening lung condition that can result from severe infections. Steroids treat pneumonia and ARDS by reducing damage the immune system can have on the lungs. The occurrence of severe pneumonia and ARDS in some COVID-19 patients led to the launch of a number of trials evaluating steroids to treat COVID-19. In June 2020, the steroid dexamethasone was the first treatment shown to reduce COVID-19 deaths. In the RECOVERY trial, outcomes were compared between approximately 2,000 hospitalized patients who received up to 10 days of dexamethasone and approximately 4,000 patients who did not. Among those who received dexamethasone, mortality at 28 days was one-third lower among patients who were mechanically ventilated and one-fifth lower among patients who required supplemental oxygen. No benefit from steroids was found in patients who did not require supplemental oxygen. These findings led the NIH to recommend using dexamethasone in COVID-19 patients who are on a ventilator or are receiving supplemental oxygen; the use of dexamethasone in mild or moderately ill COVID-19 patients is recommended against. A number of smaller trials on other steroids have also been conducted, and the pooled results of those trials suggested that dexamethasone and two other steroids were associated with reductions in mortality among critically ill COVID-19 patients. This led the World Health Organization to recommend steroids for COVID-19 patients with severe or critical illness. Steroids have now become the standard of care for critically ill COVID-19 patients. Fortunately, steroids are widely available, cheap medications that may be given intravenously or orally.
5. Tocilizumab and other IL-6 blockers
On Feb. 11, 2021, the results of an interim analysis of data from the RECOVERY trial on the efficacy of tocilizumab to treat critically ill COVID-19 patients were announced. Tocilizumab is a monoclonal antibody product that targets and blocks a human protein called interleukin 6 (IL-6), which plays a role in the inflammatory process (tocilizumab and a similar drug called sarilumab are known as “IL-6 blockers”). Given their immunomodulating properties, IL-6 blockers have been used to treat a variety of inflammatory and autoimmune diseases. In the RECOVERY trial, the outcomes for approximately 2,000 patients who received tocilizumab were compared with approximately 2,000 patients who did not; since steroids were the standard of care during the trial, 82% of participants also received a steroid such as dexamethasone. Treatment with tocilizumab significantly reduced deaths within 28 days: 596 (29%) of the patients in the tocilizumab group died compared with 694 (33%) patients in the usual care group—an absolute difference of 4%. This implies that for every 25 critically ill patients treated with tocilizumab, one additional life would be saved. However, there are conflicting and inconclusive results from other recent trials on IL-6 blockers. Although Infectious Diseases Society of America guidelines were updated to include the recommendation that COVID-19 patients who are severely or critically ill receive tocilizumab in addition to steroids, current NIH guidelines, last updated Feb, 3 2021, state that there are insufficient data to recommend either for or against the use of IL-6 blockers. WHO guidelines do not mention IL-6 blockers. Although it is possible that IL-6 blockers will become more widely recommended to treat COVID-19 patients in the future, they are expensive medications that require intravenous administration, which will limit accessibility.
Conclusion
In conclusion, there are a slowly growing number of treatments recommended for COVID-19 patients, and a lot more work to do. Steroids and other immunomodulators that tamp down overactive immune response are helping to save the lives of critically ill patients. Antiviral therapies administered early in the course of infection may help prevent severe disease in high-risk, non-hospitalized patients and may prevent death in certain critically ill hospitalized patients. Whether these treatments become more widely used may depend on building more evidence, but also on their cost, ease of administration, safety and continued efficacy as the pandemic evolves. New viral variants are not likely to affect the efficacy of immunomodulators but may reduce the efficacy of some antiviral therapies. Identifying effective, inexpensive and easy-to-administer treatments remains a priority, with many clinical trials ongoing. Even with the advances in care that have been made, outcomes are worse when hospitals are overloaded and above all, there is still no cure for COVID-19. An ideal treatment would be orally administered, safe and effective at both preventing illness after exposure and reducing the risk of severe illness and death in those who are already infected or ill. Reducing transmission is crucial to prevent severe disease and death, to stop health care systems from becoming overwhelmed and to slow down the emergence of new viral variants. As potential COVID-19 therapies are developed, tested and reviewed, we must continue to implement the 3Ws (wear a mask, wash your hands and watch your distance) to prevent COVID-19 transmission and we must support rapid and equitable vaccination.
FAQ:
What do the data say about the new Johnson & Johnson COVID-19 vaccine?
On Feb. 27, the FDA granted an EUA to the COVID-19 vaccine developed by Johnson and Johnson (J&J), which is 66% effective against moderate to severe COVID-19 illness. The J&J vaccine was 85% effective at preventing severe COVID-19 illness (from 28-days after vaccination) and 100% effective at preventing COVID-19-related hospitalization and death (28-days after vaccination). The vaccine also reduced asymptomatic infection, which means that it could help stop transmission of COVID-19. Unlike the Pfizer and Moderna vaccines previously available in the U.S. under EUA, the J&J vaccine uses an adenovirus-vector platform (rather than mRNA) and requires only one dose; it is authorized for use in adults aged 18 or older. Because J&J and mRNA vaccines are safe and highly effective at preventing severe disease, hospitalization and death, experts recommend those eligible for vaccination take any available EUA-authorized vaccine.
- The study was a randomized, placebo-controlled trial including approximately 40,000 participants in the U.S. (47%), South Africa (13%), Brazil (17%) and 5 other Latin American Countries (23%). We reported previously on an earlier release of data from this study.
- The vaccine was similarly efficacious across age, race and ethnic groups, although lower efficacy was observed among participants aged 60 years and older with comorbidities, compared with the overall population; however, the follow-up period was shorter in this group and more data are needed to provide a reliable estimate.
- The vaccine appears to offer protection against asymptomatic infection, which means that it may help to stop the spread of COVID-19. Among participants with a positive PCR or antibody test for COVID-19 and no history of symptoms, vaccine efficacy was 74% (95% CI: 46.8; 88.4).
- Efficacy of the vaccine appeared to be higher in the U.S. (72%) compared to South Africa (64%), although the efficacy against severe disease was similar in both locations (86% in the U.S., 82% in South Africa).
- Sequencing data were available for 72% of cases included in the study. Among cases with sequencing data, 96% of U.S. cases were Wuhan-H1 variant D614G, whereas 95% of cases in South Africa were variant B.1.351 and 69% of cases in Brazil were variant P.2. There were no cases identified as B.1.1.7 or P.1 lineages as of Feb. 12, 2021.
- As of Feb. 5, 2021, there were 7 COVID-19-related deaths reported in the study, all of which occurred in the placebo group and were in study sites in South Africa.
- Side effects reported after vaccination were predominantly mild and moderate; the most common side effects were injection site pain (49%), headache (39%), fatigue (38%) and myalgia (33%). Non-fatal serious adverse events, excluding those due to COVID-19, were infrequent and balanced between the vaccine and placebo groups with respect to rates and types of events (0.4% in both groups).
- J&J is continuing to study the efficacy of a two-dose vaccine regimen.
Weekly Research Highlights
Covid-19 deaths in Africa: prospective systematic postmortem surveillance study
(BMJ February 2021)
- Researchers conducted PCR testing for SARS-CoV-2 on a systematic random sample of deceased people of all ages at the University Teaching Hospital morgue in Lusaka, Zambia, which registers at least 80% of all deaths in the city. Information on comorbidities and underlying risk factors were collected either from medical records or interviews with families of the deceased. During June 15- Oct. 1, 2020, 372 deceased participants were enrolled, representing 10% of all deaths registered at the morgue.
- Deaths with COVID-19 occurred throughout the entire study period with notable week-to-week variation. The highest proportion of deaths with COVID-19 occurred in late July and early August, corresponding to an overall increase in cases in Zambia.
- The median age of death was 48 (interquartile range 36-72 years) with two-thirds of all deaths clustered among people aged 20-59 years.
- The most common comorbidities among people who died with COVID-19 were tuberculosis (31%), hypertension (27%) and HIV/AIDS (23%). With the exception of hypertension, COVID-19-associated comorbidities that typically occur in aging populations were not common in this Zambian study population.
- Limitations: The study was conducted over a short span of months, in one city in one African country, and may not be representative of the entire African context. Given the variable distribution of deaths over time, patterns seen in this study will likely not represent trends over a longer period of time, justifying the need for ongoing surveillance. Another limitation is that cause of death could not be inferred, and it is not clear whether participants died from COVID-19 or, potentially, had mild infections with SARS-CoV-2 that did not directly contribute to death. Further, assessment of comorbidities and risk factors was limited by the availability and accuracy of data.
Early rate reductions of SARS-CoV-2 infection and COVID-19 in BNT162b2 vaccine recipients
(The Lancet, February 2021)
- Researchers analyzed data from Israel’s largest hospital. They conducted a retrospective cohort analysis of 9,109 vaccine-eligible HCWs. These HCWs conducted daily symptom reporting and had access to same-day SAR-CoV-2 PCR testing. Data were analyzed during Dec. 19, 2020-Jan. 24, 2021, by which time 79% of HCWs had received a first dose and 66% had received the second dose. During this time, 170 HCWs experienced a SARS-CoV-2 infection, 99 of whom reported symptomatic disease.
- Among the 170 HCWs with SARS-CoV-2 infection, 89 cases occurred among unvaccinated HCWs (7.4 per 10,000 person-days), 55 occurred among HCWs 1-14 days after their first dose (5.5 per 10,000 person-days) and 26 occurred among HCWs 15-28 days after their first dose (3.0 per 10,000 person-days). After adjusting for community exposure rates for COVID-19, individuals had a 75% lower rate of SARS-CoV-2 infection 15-28 days after their first dose compared to unvaccinated HCWs.
- Of the 99 HCWs experiencing symptomatic COVID-19, 60 occurred among unvaccinated HCW (5.0 per 10,000 person-days), 28 occurred 1-14 days after the first dose (2.8 per 10,000 person-days) and 11 occurred 15-28 days after the first dose (1.2 per 10,000 person-days). In an adjusted analysis, HCWs 15-28 days after their first dose had an 85% lower rate of COVID-19 compared to unvaccinated HCWs.
- Limitations of this analysis include the fact that it was an observational study and not a randomized control trial meaning that differences between groups being compared could affect the results. Additionally, HCWs were not routinely tested for SARS-CoV-2, so some asymptomatic cases may not have been detected. Vaccine effectiveness among HCWs may differ from that of the general population due to HCWs potentially experiencing higher levels of exposure to SARS-CoV-2. The study also does not address the long-term effectiveness of a single dose of vaccine.
Suggested citation: Cash-Goldwasser S, Jones SA, Cobb L, Bochner A, Subramaniam H, Bradford E and Frieden TR. In-Depth COVID-19 Science Review February 22 – March 2, 2021. Resolve to Save Lives. 2021 March 2. Available from https://preventepidemics.org/covid19/science/review/





